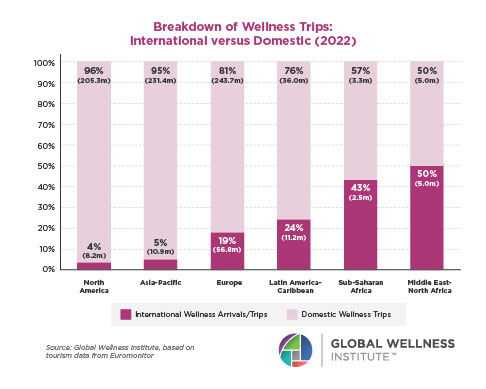
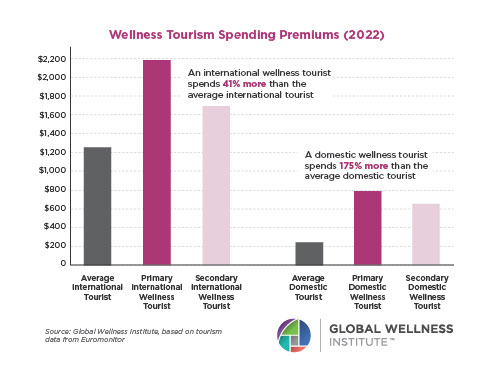
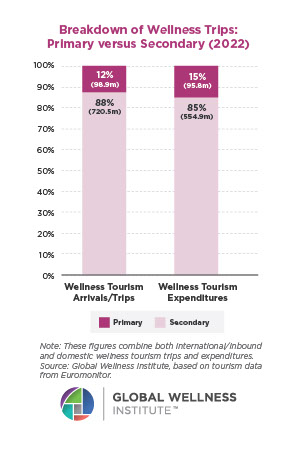
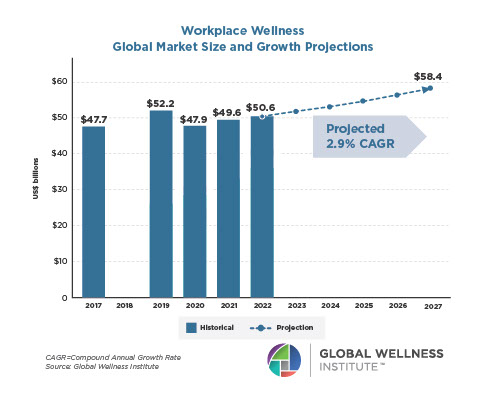
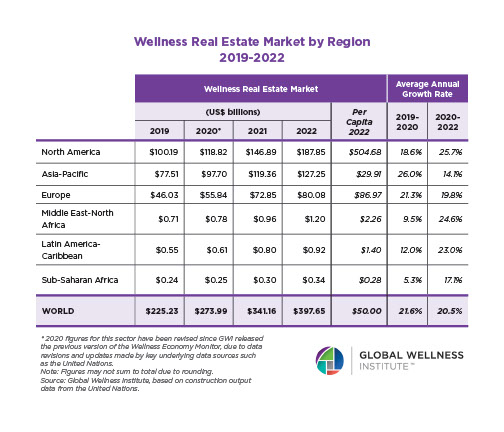
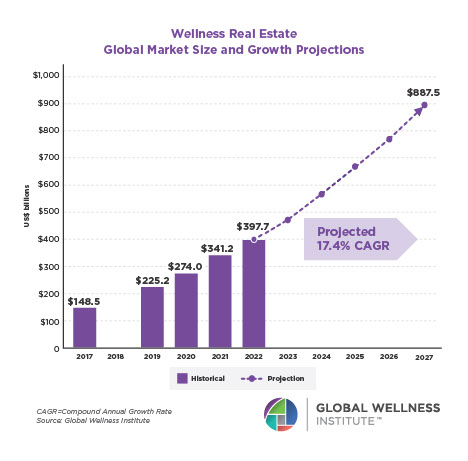
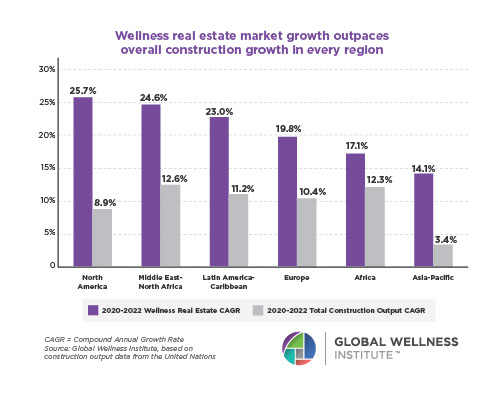
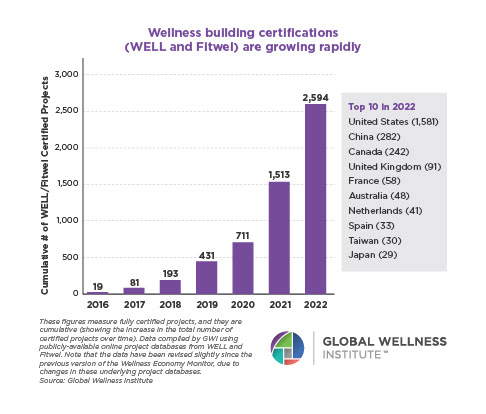
Why, Y chromosome, why?
The Y chromosome is a developmental switch. A single gene on the Y chromosome — SRY (Sex-determining Region Y) — initiates the differentiation of the bipotential gonad into a testis. Without functional SRY, the gonad develops as an ovary regardless of karyotype; with SRY translocated to an X chromosome, XX individuals develop as phenotypic males (Wilson & Davies, 2007). Once SRY has thrown the switch, the genetic variation that shapes behavioral differences among men lives elsewhere — on the X chromosome, on autosomes, and in the epigenetic modifications that accumulate across development. The Avon Longitudinal Study of Parents and Children (n = 4,788 males with Y chromosome haplogroup data) found no association between Y haplogroups and any childhood behavioral or psychiatric trait assessed — spanning ADHD/attention-activity, conduct problems, emotional symptoms, autistic traits, and psychotic experiences measured at ages 7 through 18 — despite the majority of these traits showing significant sex differences in the same cohort (Howe et al., 2017). In over 1,000 young adult white men from the UK general population, Y chromosome haplogroup I — a lineage carrying approximately 50% elevated coronary artery disease risk — showed no association with any of the four Buss-Perry aggression subscales (physical, verbal, anger, hostility) or with circulating androgen or estrogen levels (Bloomer et al., 2014). At the broadest scale, only 9 of the 447,939 genome-wide-significant associations accumulated in the NHGRI-EBI GWAS Catalog through late 2022 map to the Y chromosome (Sun et al., 2023) — a significantly low yield across decades of behavioral, psychiatric, and medical GWAS, particularly when compared to X-linked traits (as an example, see Luciano et al., 2021). Thus, common Y-linked genetic variation does not appear to explain why men differ from one another in temperament, aggression, prosociality, or other behavioral phenotypes.
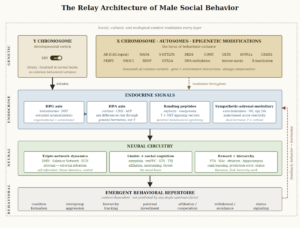
SRY does have one direct effect on the brain that bypasses the testicular pathway. SRY protein is expressed in catecholamine-rich regions of the adult male brain — the substantia nigra pars compacta, ventral tegmental area, locus coeruleus, and hypothalamus — where it participates in the regulation of dopamine and noradrenaline synthesis (Lee & Harley, 2012; Czech et al., 2012). SRY knockdown in rat substantia nigra reduces tyrosine hydroxylase-positive neurons and impairs motor performance (Dewing et al., 2006); SRY overexpression in human neuroblastoma cells increases catecholamine synthesis enzymes and extracellular dopamine (Czech et al., 2012). This is a confirmed Y-linked neural mechanism independent of circulating testosterone, and it may contribute to male-biased susceptibility to Parkinson’s disease and other catecholamine-related disorders. As a contributor to male social behavior, however, its scope may be narrow: SRY may bias the sympathetic stress response toward fight-or-flight activation, but not toward any specific behavioral outcome.
From the moment that SRY initiates the testes differentiation in the fetus, their function is endocrine: they produce testosterone, and the foremost target of testosterone in embryonic development is the maturing male brain. Prenatal testosterone, acting through androgen receptors whose density and distribution are encoded on the X chromosome and autosomes, organizes the neural architecture that will later be activated by pubertal and adult hormones — shaping amygdala volume, hypothalamic nuclei, and the receptor landscape across cortical and subcortical regions (Auyeung et al., 2013; Hines, 2020). In humans, this masculinization operates primarily through direct androgen receptor action (Wilson & Davies, 2007). The organizational-activational framework — first articulated by Phoenix et al. (1959) and refined over six decades — holds that prenatal hormones organize male-type neural circuits that adult hormones later activate.
In the brain, testosterone’s behavioral effects are mediated by the expression of numerous other genes. The androgen receptor gene (AR, Xq11-12) is X-linked, meaning each man inherits a single allele from his mother. A polymorphic CAG repeat in the AR gene modulates receptor sensitivity — shorter repeats produce greater transcriptional activity and greater cellular response to testosterone — and the behavioral consequences are ecology-dependent: among Hadza foragers, shorter repeats predict greater aggression and higher reproductive output (Butovskaya et al., 2015); in Western populations, the same association is frequently null (Valenzuela et al., 2022). Monoamine oxidase A (MAOA, Xp11.3), also X-linked, encodes the enzyme that degrades serotonin, dopamine, and norepinephrine. The low-activity variant (MAOA-L) produces elevated monoamine levels in limbic regions, lowering the threshold for reactive aggression — but only in the presence of childhood maltreatment (Caspi et al., 2002; Byrd & Manuck, 2014, meta-analysis across 20 male samples, failsafe N > 93). The MAOA × maltreatment interaction is specific to reactive, impulsive aggression; it does not predict proactive, planned aggression, which operates through a different neural pathway entirely (Kolla & Bortolato, 2020).
The resulting genetic architecture of masculinity has a counterintuitive structure. The Y chromosome initiates male development but does not encode behavioral variation. The genes that modulate how any given man responds to testosterone — the androgen receptor, MAOA, and the oxytocin receptor — sit on the X chromosome and autosomes rather than the Y, and their behavioral expression is shaped by developmental context and ecological conditions (as the AR and MAOA evidence above shows). Masculinity is assembled primarily from genetic components that are neither deterministic nor male-specific. What converts this genetic predisposition into a behavioral phenotype is the endocrine system — the hormonal architecture that organizes and activates male neural circuits across the lifespan. Before turning to how male neural circuits are activated today, the next essay traces how they evolved.
References
Auyeung, B., Lombardo, M. V., & Baron-Cohen, S. (2013). Prenatal and postnatal hormone effects on the human brain and cognition. Pflügers Archiv – European Journal of Physiology, 465(5), 557–571. https://doi.org/10.1007/s00424-013-1268-2
Bloomer, L. D. S., Nelson, C. P., Denniff, M., Christofidou, P., Debiec, R., Thompson, J., Zukowska-Szczechowska, E., Samani, N. J., Charchar, F. J., & Tomaszewski, M. (2014). Coronary artery disease predisposing haplogroup I of the Y chromosome, aggression and sex steroids — Genetic association analysis. Atherosclerosis, 233(1), 160–164. https://doi.org/10.1016/j.atherosclerosis.2013.12.012
Butovskaya, M. L., Lazebny, O. E., Vasilyev, V. A., Dronova, D. A., Karelin, D. V., Mabulla, A. Z. P., Shibalev, D. V., & Ryskov, A. P. (2015). Androgen receptor gene polymorphism, aggression, and reproduction in Tanzanian foragers and pastoralists. PLOS ONE, 10(8), e0136208. https://doi.org/10.1371/journal.pone.0136208
Byrd, A. L., & Manuck, S. B. (2014). MAOA, childhood maltreatment, and antisocial behavior: Meta-analysis of a gene–environment interaction. Biological Psychiatry, 75(1), 9–17. https://doi.org/10.1016/j.biopsych.2013.05.004
Caspi, A., McClay, J., Moffitt, T. E., Mill, J., Martin, J., Craig, I. W., Taylor, A., & Poulton, R. (2002). Role of genotype in the cycle of violence in maltreated children. Science, 297(5582), 851–854. https://doi.org/10.1126/science.1072290
Czech, D. P., Lee, J., Sim, H., Parish, C. L., Vilain, E., & Harley, V. R. (2012). The human testis-determining factor SRY localizes in midbrain dopamine neurons and regulates multiple components of catecholamine synthesis and metabolism. Journal of Neurochemistry, 122(2), 260–271. https://doi.org/10.1111/j.1471-4159.2012.07782.x
Dewing, P., Chiang, C. W. K., Sinchak, K., Sim, H., Fernagut, P.-O., Kelly, S., Chesselet, M.-F., Micevych, P. E., Albrecht, K. H., Harley, V. R., & Vilain, E. (2006). Direct regulation of adult brain function by the male-specific factor SRY. Current Biology, 16(4), 415–420. https://doi.org/10.1016/j.cub.2006.01.017
Hines, M. (2020). Neuroscience and sex/gender: Looking back and forward. Journal of Neuroscience, 40(1), 37–43. https://doi.org/10.1523/JNEUROSCI.0750-19.2019
Howe, L. J., Erzurumluoglu, A. M., Davey Smith, G., Rodriguez, S., & Stergiakouli, E. (2017). Y chromosome, mitochondrial DNA and childhood behavioural traits. Scientific Reports, 7(1), 11655. https://doi.org/10.1038/s41598-017-10871-4
Kolla, N. J., & Bortolato, M. (2020). The role of monoamine oxidase A in the neurobiology of aggressive, antisocial, and violent behavior: A tale of mice and men. Progress in Neurobiology, 194, 101875. https://doi.org/10.1016/j.pneurobio.2020.101875
Lee, J., & Harley, V. R. (2012). The male fight–flight response: A result of SRY regulation of catecholamines? BioEssays, 34(6), 454–457. https://doi.org/10.1002/bies.201100159
Luciano, M., Davies, G., Summers, K. M., Hill, W. D., Hayward, C., Liewald, D. C., Porteous, D. J., Hagenaars, S. P., Harris, S. E., McIntosh, A. M., & Deary, I. J. (2021). The influence of X chromosome variants on trait neuroticism. Molecular Psychiatry, 26(2), 483–491. https://doi.org/10.1038/s41380-019-0388-2
Phoenix, C. H., Goy, R. W., Gerall, A. A., & Young, W. C. (1959). Organizing action of prenatally administered testosterone propionate on the tissues mediating mating behavior in the female guinea pig. Endocrinology, 65(3), 369–382. https://doi.org/10.1210/endo-65-3-369
Sun, L., Wang, Z., Lu, T., Manolio, T. A., & Paterson, A. D. (2023). eXclusionarY: 10 years later, where are the sex chromosomes in GWASs? American Journal of Human Genetics, 110(6), 903–912. https://doi.org/10.1016/j.ajhg.2023.04.009
Valenzuela, N. T., Ruiz-Pérez, I., Rodríguez-Sickert, C., Polo, P., Muñoz-Reyes, J. A., Yeste-Lizán, A., & Pita, M. (2022). The relationship between androgen receptor gene polymorphism, aggression and social status in young men and women. Behavioral Sciences, 12(2), 42. https://doi.org/10.3390/bs12020042
Wilson, C. A., & Davies, D. C. (2007). The control of sexual differentiation of the reproductive system and brain. Reproduction, 133(2), 331–359. https://doi.org/10.1530/REP-06-0078