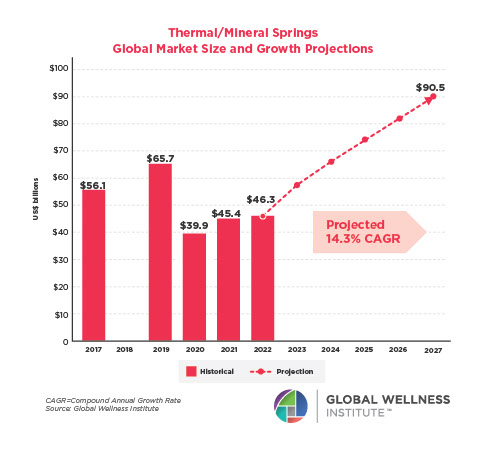
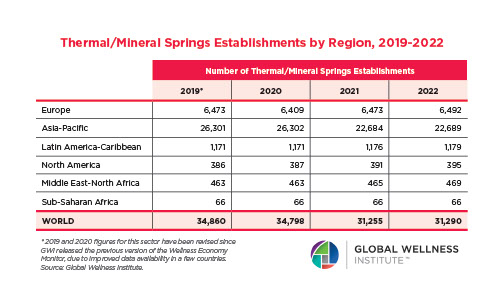
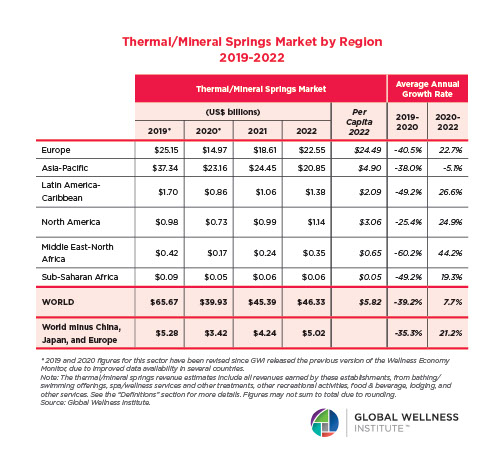
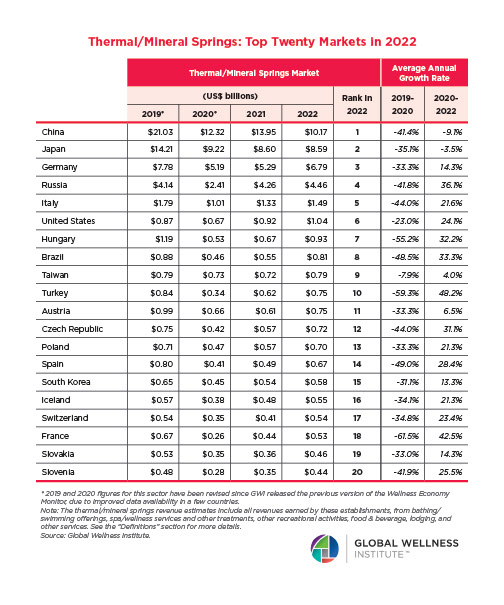
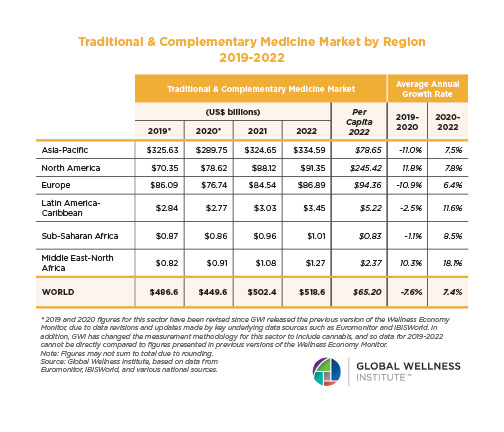
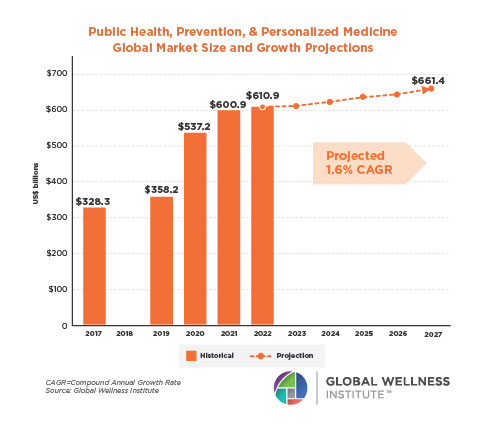
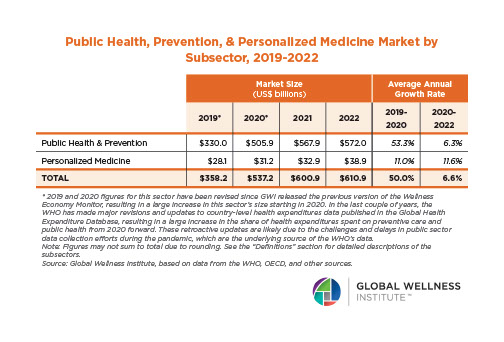
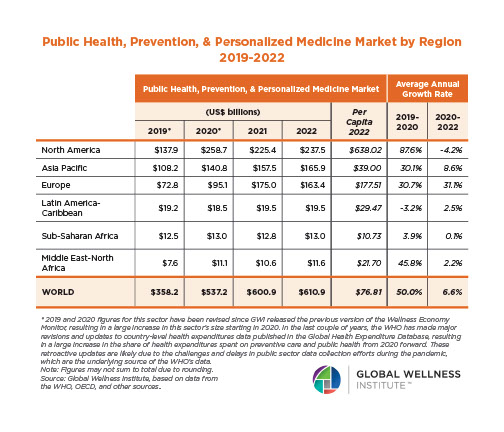
The Male Role in a Group
The Male Role Across Social Mammals
The male role in a mammalian group is assembled through a developmental pathway initiated by the Y chromosome. Sry triggers testis formation, the fetal testes secrete testosterone, and the hormone acts during a perinatal sensitive period to organize the developing brain along male-typical lines (Phoenix et al., 1959; Arnold, 2009). In primates, androgens acting directly on androgen receptors carry out this masculinization; in rodents, the active hormone is estradiol aromatized from testosterone within neurons themselves (Wallen, 2005; Thornton et al., 2009). Female rhesus macaques exposed to androgens late in gestation show masculinized rough-and-tumble play despite unmasculinized genitalia, separating the behavioral effect from any social response to the infant’s appearance (Wallen, 2005). Juvenile males across mammalian species engage in rough-and-tumble play at higher rates and with greater intensity than females, with the pattern documented from rodents through non-human primates to humans (Auger & Olesen, 2009).
The brain this pathway produces evolved in groups, and the male role within those groups exhibits consistent patterns across social mammals that predate the primate lineage. In a comparative phylogenetic analysis of 72 group-living mammalian species — spanning herbivores (feral horses), omnivores (white-faced capuchins), and carnivorans (grey wolves) — males more often led intergroup conflict, whereas females more often led collective movements toward food, water, and safety (Smith et al., 2022). The sex bias in intergroup conflict was not predicted by differences in body size between genders — many species with male-biased conflict participation show minimal body size differences between sexes — but, rather, by the differential cost-benefit structure of mammalian reproduction. Male fitness is limited primarily by access to mates, and intergroup conflict provides opportunities for mate acquisition, territory expansion, and defense of reproductive partners. Female fitness is limited primarily by access to food and the energetic demands of gestation and lactation, and collective movements toward resources directly serve these needs. This pattern has persisted across primates and non-primates, suggesting that the sex-typed division of group labor is older than the primate lineage itself.
Within this inherited architecture, male aggression varies in form with the target — outgroup rival, intragroup competitor, or norm violator — and affiliative approach shifts toward or away from the group depending on rank and stress. Cooperation and competition within the group are states that change with whether the outgroup is salient. None of this functions without a continuously updated read of the group: who outranks whom, who is allied with whom, and where ingroup and outgroup lines fall at a given moment. The behavioral repertoire and the social-cognitive machinery that orients it are components of a single system, shaped by the fitness demands of our group-living ancestors.
The Behavioral Dimensions of Male Group Life
The behaviors that males exhibit within groups can be deconvolved into separable dimensions, each with different neural pathways, hormonal regulation, and evolutionary histories. The first deconvolution distinguishes reactive aggression from proactive aggression. Wrangham (2018) assembled the evidence that these two types of aggression have different neural pathways (reactive: amygdala-driven with impaired prefrontal regulation; proactive: prefrontal-mediated and goal-directed), different developmental trajectories, and — in the human lineage — opposite evolutionary histories. Humans show reduced reactive aggression relative to chimpanzees, consistent with the self-domestication hypothesis and supported by craniofacial feminization in the fossil record over the past 200,000 years (Cieri et al., 2014). Humans simultaneously show elevated proactive aggression — the capacity for planned, coalitional violence that requires cooperation among attackers and is shared with chimpanzees but absent in bonobos. One proposed selection mechanism is coordinated capital punishment of aggressive norm violators: in Boehm’s (2012) model, ancestral groups that executed selfish aggressors would have created selection pressure against reactive aggression while preserving and possibly enhancing the proactive capacity required for the executions themselves. This remains a hypothesis, but it is consistent with the phylogenetic pattern, and it offers one account of how two types of aggression could have been shaped by different selection pressures operating within the same species.
The second dimension separates affiliative approach from stress-withdrawal, describing how males vary in how readily they seek social contact versus withdraw from it, particularly under stress. In a field study of real-world competition, Sherman et al. (2017) found a sex-crossover effect in which rising cortisol after defeat predicted increased affiliation in women but decreased affiliation in men — a finding discussed in full in a following essay on endocrine signaling. The underlying mechanism has a plausible molecular basis: cortisol has been shown to block testosterone’s effects on status-seeking and social approach behavior through glucocorticoid antagonism of androgen receptor function, pushing the high-cortisol male toward disengagement rather than re-engagement (Mehta & Josephs, 2010). Real-world evidence lends further support: in marriages, husbands under stress are substantially more likely than their wives to withdraw and seek interpersonal distance (Christensen & Heavey, 1990; Gottman & Levenson, 1988), and a broader meta-analysis found that the largest sex difference in stress coping is women’s greater tendency to seek emotional support from others (Tamres et al., 2002). Under experimental stress, men also provide lower-quality social support to relationship partners than women do (Bodenmann et al., 2015). The male-typical stress response is more specifically characterized as affiliative withdrawal than as undifferentiated fight-or-flight — a pattern consistent with the ecology of dominance hierarchies, where re-engaging a superior after defeat risks escalation. This dimension is partially independent of aggression: a withdrawn male is not hostile toward his group; he is simply not engaging with it.
The third dimension is cooperative versus competitive orientation toward the ingroup. Muñoz-Reyes et al. (2020) tested this directly by placing young men in intergroup competition and measuring both aggression and cooperation simultaneously. Intergroup conflict increased both: aggression toward the outgroup and cooperation toward the ingroup rose in tandem, and the men who were most aggressive toward outgroup rivals were the most cooperative with ingroup allies. The effect was modulated by pubertal testosterone proxies — muscle mass predicted both behaviors under outgroup threat — suggesting that the same androgenic physiology that equips males for physical contest also facilitates ingroup coordination when both are activated by intergroup challenge.
Coalitional cooperation also shapes dominance trajectories within a group. Neumann et al. (2022) tracked 128 coalition events among 28 wild male crested macaques (Macaca nigra) in the Tangkoko Nature Reserve over two years, combining behavioral observation, Elo-rating to quantify rank dynamics, and genetic paternity analysis of 19 infants conceived during the study. Males who participated in coalitions achieved higher future ranks than expected; males targeted by coalitions dropped lower than expected. The magnitude of the rank shift depended on coalition configuration. In all-up coalitions — where every participant ranked below the target at the time of the event — the rank effect was largest, and was greatest when the combined strength of the participants was low relative to the target. The participants in the riskiest coalitions profited most. Bridging coalitions (with at least one participant above and at least one below the target) and all-down coalitions (all participants outranking the target) produced smaller effects, though all-down coalitions — which the coalition-formation literature has traditionally treated as non-profitable — measurably sustained rank trajectories that would otherwise have drifted downward. Paternity analysis linked these rank changes to reproductive outcomes: higher-ranking males around the date of conception were more likely to sire infants, connecting coalitional participation to fitness through a dominance-trajectory pathway.
Cooperation and aggression are context-dependent outputs of the same neuroendocrine substrate. The target of cooperation can be a coalitional ally, with the aggression directed at either an outgroup rival or an intragroup rival, and it is the identity of that target — not the presence or absence of cooperation — that determines which behavioral pathway activates.
Dominance and Subordination as Communicative Acts
Participating in a group depends on reading it accurately. A male needs to know who outranks whom, who is allied with whom, and where ingroup and outgroup lines fall at any given moment — assessments that can shift across contexts and over time. Two systems perform this work: a signaling channel that regulates rank without repeated physical contest, and a categorization system that tracks coalitional affiliation.
Among primates, dominance and subordination signals include postural expansion or contraction, direct or averted gaze, vocal pitch lowering or elevation, and facial displays, such as the silent bared-teeth gesture in macaques. These signals communicate the state of a dyadic relationship without either party needing to fight to establish it. Subordination is associated with cortisol elevation, postural contraction, and gaze aversion across primates, while dominance is associated with testosterone elevation, postural expansion, and direct gaze. The signaling system converts what would otherwise require constant physical testing — who outranks whom — into a low-cost information exchange that stabilizes the hierarchy and reduces within-group violence.
De Waal’s reconciliation research (de Waal & van Roosmalen, 1979; de Waal, 2000) documented that chimpanzees kiss and embrace after fights, and that these post-conflict affiliative contacts serve to repair social relationships disrupted by aggression. The finding has been replicated across more than a dozen primate species, including rhesus macaques, stump-tailed macaques, bonobos, and capuchins, and has been extended to non-primate species including wolves, domestic dogs, horses, and spotted hyenas (Aureli & de Waal, 2000).
Subordination signals in pigtailed macaques have context-dependent meaning (Flack & de Waal, 2007). When the silent bared-teeth display is produced during conflict, it communicates immediate submission — a tactical concession in the current encounter. When the same display is produced in peaceful contexts, it communicates subordination — a commitment to the long-term pattern of the relationship, reducing the receiver’s uncertainty about the sender’s future behavior. In rhesus macaques, dyads that exchanged peaceful subordination signals had less frequent severe aggression and higher rates of grooming than dyads that signaled only during conflict or not at all (Beisner & McCowan, 2014). While withdrawal removes the individual from the social field entirely, subordination signaling maintains the individual’s engagement with the group while communicating acceptance of lower rank. The signal reduces the probability of attack from the dominant and permits the subordinate to access the benefits of group membership — food, protection, mating opportunities — that withdrawal would forfeit.
These signals are phylogenetically conserved communicative acts with identifiable physiological signatures. As an example, the evolutionary trajectory of the silent bared-teeth display goes roughly like this: in despotic species with steep hierarchies (rhesus macaques), the display stays tightly bound to its subordination function — only subordinates produce it, only dominants receive it. In more socially tolerant species (Tonkean macaques, stumptailed macaques), the display becomes bidirectional — both dominants and subordinates produce it, and it takes on an affiliative, reassurance function closer to what we, humans, recognize as a smile. In chimpanzees and bonobos, the display is used across rank directions and in explicitly affiliative contexts — greeting, reconciliation, play solicitation. The muscles are the same (zygomaticus major, orbicularis oculi), the facial configuration is recognizably the same, but the social meaning has broadened from “I submit” to “I am not a threat” to something approaching “I am positively disposed toward you” (Preuschoft & van Hooff, 1997).
Human smiling appears to sit at the far end of this gradient: our display of this signal has been fully decoupled from its original subordination context and serves a broader range of social functions (including affiliation, appeasement, greeting, reward, deception). Yet, traces of the original function persist: people smile more at higher-status individuals than at lower-status ones (Ketelaar et al., 2012), and subordinates smile more than dominants in workplace interactions (Méhu & Dunbar, 2008), which may be our ancestral primate pattern showing through.
Recognizing Others: Allies and Kin
Alongside the signaling system that handles rank, a second system handles alliances. Kurzban et al. (2001) demonstrated that encoding by coalitional affiliation is a core feature of person representation — subjects automatically tracked which individuals belonged to which alliance in a novel social scenario. When coalitional cues were made salient, categorization by race — previously thought to be automatic and mandatory — dropped to statistical nonsignificance after less than four minutes of exposure. Categorization by sex, by contrast, was unaffected. The result supports the hypothesis that the human mind contains evolved machinery for detecting coalitional alliances, and that racial categorization is a byproduct of this machinery rather than a primary adaptation. Our coalitional computation system operates on any cue that reliably predicts who will cooperate with whom: shared appearance, shared insignia, behavioral coordination, verbal declarations of allegiance.
The mechanisms by which specific individuals are categorized as kin draw on three converging systems identified across vertebrates (Waldman, 1987; Holmes & Sherman, 1982; Mateo, 2015). Spatial association — co-residence during a critical developmental window — is the most phylogenetically widespread: organisms that grow up together treat each other as kin regardless of genetic relatedness, a mechanism confirmed experimentally in humans (Lieberman et al., 2007). Phenotypic matching — recognizing individuals who share heritable features such as facial geometry, voice quality, and body odor — operates through a self-referential template: individuals compare a target’s phenotype to their own or to familiar kin, and the degree of overlap modulates social behavior (DeBruine, 2002; Bressan & Zucchi, 2009). Familiarity-based individual recognition, built through repeated interaction and encoded in hippocampal memory systems, provides the third channel and permits discrimination among known individuals within a kinship class.
For males, the father occupies a specific position in this categorization architecture. In biparental species — a minority among mammals but characteristic of the human lineage — the father is the first non-maternal adult male that the offspring’s recognition systems encounter during the critical period in which social categories are being assembled. Across the pair-bonding mammals in which paternal care has evolved, fathers who engage in direct caregiving during this window produce sons whose affiliative behavioral repertoire differs measurably from that of sons raised without paternal contact. The Gleason and Marler (2013) California mouse (Peromyscus californicus) cross-fostering finding — that sons of attentive fathers become attentive fathers through non-genomic transmission that tracks rearing rather than biological paternity — is the clearest animal model for this effect and is discussed in detail in the Endocrine essay. In humans, the analogous process operates through biobehavioral synchrony (Feldman, 2012): father-infant interaction during the first years of life, characterized by the physically stimulatory, rough-and-tumble play style that distinguishes paternal from maternal interaction across cultures, sets parameters of the affiliative system that the son’s endocrine system will later modulate.
The Architecture Expects a Group
The behavioral dimensions described here — reactive and proactive aggression, affiliative approach versus stress-withdrawal, cooperative versus competitive orientation, dominance and subordination signaling, kin and alliance categorization — are not independent modules operating in isolation. They are components of an integrated system that evolved to function inside a social group. Reactive aggression is held in check by the subordination signaling system. Proactive aggression is channeled by the ingroup/outgroup categorization system toward external threats rather than internal rivals. Affiliative approach is calibrated by the oxytocin system, which is itself set by early caregiving experience. Cooperative behavior is gated by the testosterone-cortisol interaction, which adjusts the balance between competitive and cooperative strategies as a function of whether the social context provides outgroup threat or ingroup stability.
| Dimension | Neural pathway | Endocrine modulator | Key evidence |
|---|---|---|---|
| Reactive vs. proactive aggression | Amygdala-driven (reactive) vs. PFC-mediated (proactive) | Serotonin / MAOA (reactive); dopamine (proactive) | Wrangham, 2018; self-domestication hypothesis |
| Affiliative approach vs. stress-withdrawal | Oxytocin system (approach); HPA gating (withdrawal) | Cortisol suppresses affiliation in men | Sex-crossover: cortisol ↑ → affiliation ↓ in men, ↑ in women (Sherman et al., 2017) |
| Cooperative vs. competitive orientation | DLPFC (cooperation); ventral striatum (competition) | Testosterone + outgroup threat = both increase | Aggression and cooperation rise simultaneously under intergroup threat (Muñoz-Reyes et al., 2020) |
| Dominance / subordination signaling | vmPFC (rank representation); amygdala (status evaluation) | Testosterone (status-seeking); cortisol gates expression | Zink et al., 2008; Kumaran et al., 2012 |
| Kin and alliance categorization | mPFC, fusiform face area, TPJ | Oxytocin (ingroup bias) | Categorization tracks alliance, not phenotype (Kurzban et al., 2001) |
The following essays trace this system through its neural circuitry, its endocrine regulation, its developmental assembly, and the consequences of the environmental mismatch that characterizes modern men’s ecology.
References
Arnold, A. P. (2009). The organizational–activational hypothesis as the foundation for a unified theory of sexual differentiation of all mammalian tissues. Hormones and Behavior, 55(5), 570–578. https://doi.org/10.1016/j.yhbeh.2009.03.011
Auger, A. P., & Olesen, K. M. (2009). Brain sex differences and the organisation of juvenile social play behaviour. Journal of Neuroendocrinology, 21(6), 519–525. https://doi.org/10.1111/j.1365-2826.2009.01871.x
Aureli, F., & de Waal, F. B. M. (Eds.). (2000). Natural conflict resolution. University of California Press.
Beisner, B. A., & McCowan, B. (2014). Signaling context modulates social function of silent bared-teeth displays in rhesus macaques (Macaca mulatta). American Journal of Primatology, 76(2), 111–121. https://doi.org/10.1002/ajp.22214
Bodenmann, G., Meuwly, N., Germann, J., Nussbeck, F. W., Heinrichs, M., & Bradbury, T. N. (2015). Effects of stress on the social support provided by men and women in intimate relationships. Psychological Science, 26(10), 1584–1594. https://doi.org/10.1177/0956797615594616
Boehm, C. (2012). Moral origins: The evolution of virtue, altruism, and shame. Basic Books.
Bressan, P., & Zucchi, G. (2009). Human kin recognition is self- rather than family-referential. Biology Letters, 5(3), 336–338. https://doi.org/10.1098/rsbl.2008.0789
Christensen, A., & Heavey, C. L. (1990). Gender and social structure in the demand/withdraw pattern of marital conflict. Journal of Personality and Social Psychology, 59(1), 73–81. https://doi.org/10.1037/0022-3514.59.1.73
Cieri, R. L., Churchill, S. E., Franciscus, R. G., Tan, J., & Hare, B. (2014). Craniofacial feminization, social tolerance, and the origins of behavioral modernity. Current Anthropology, 55(4), 419–443. https://doi.org/10.1086/677209
DeBruine, L. M. (2002). Facial resemblance enhances trust. Proceedings of the Royal Society B: Biological Sciences, 269(1498), 1307–1312. https://doi.org/10.1098/rspb.2002.2034
de Waal, F. B. M. (2000). Primates — a natural heritage of conflict resolution. Science, 289(5479), 586–590. https://doi.org/10.1126/science.289.5479.586
de Waal, F. B. M., & van Roosmalen, A. (1979). Reconciliation and consolation among chimpanzees. Behavioral Ecology and Sociobiology, 5(1), 55–66. https://doi.org/10.1007/BF00302695
Feldman, R. (2012). Bio-behavioral synchrony: A model for integrating biological and microsocial behavioral processes in the study of parenting. Parenting: Science and Practice, 12(2–3), 154–164. https://doi.org/10.1080/15295192.2012.683342
Flack, J. C., & de Waal, F. B. M. (2007). Context modulates signal meaning in primate communication. Proceedings of the National Academy of Sciences, 104(5), 1581–1586. https://doi.org/10.1073/pnas.0603565104
Gleason, E. D., & Marler, C. A. (2013). Non-genomic transmission of paternal behaviour between fathers and sons in the monogamous and biparental California mouse. Proceedings of the Royal Society B: Biological Sciences, 280(1763), 20130824. https://doi.org/10.1098/rspb.2013.0824
Gottman, J. M., & Levenson, R. W. (1988). The social psychophysiology of marriage. In P. Noller & M. A. Fitzpatrick (Eds.), Perspectives on marital interaction (pp. 182–200). Multilingual Matters.
Holmes, W. G., & Sherman, P. W. (1982). The ontogeny of kin recognition in two species of ground squirrels. American Zoologist, 22(3), 491–517. https://doi.org/10.1093/icb/22.3.491
Ketelaar, T., Koenig, B. L., Gambacorta, D., Dolgov, I., Hor, D., Zarzosa, J., Luna-Nevarez, C., Klungle, M., & Wells, L. (2012). Smiles as signals of lower status in football players and fashion models: Evidence that smiles are associated with lower dominance and lower prestige. Evolutionary Psychology, 10(3), 371–397. https://doi.org/10.1177/147470491201000301
Kurzban, R., Tooby, J., & Cosmides, L. (2001). Can race be erased? Coalitional computation and social categorization. Proceedings of the National Academy of Sciences, 98(26), 15387–15392. https://doi.org/10.1073/pnas.251541498
Lieberman, D., Tooby, J., & Cosmides, L. (2007). The architecture of human kin detection. Nature, 445(7129), 727–731. https://doi.org/10.1038/nature05510
Mateo, J. M. (2015). Perspectives: Hamilton’s legacy: Mechanisms of kin recognition in humans. Ethology, 121(5), 419–427. https://doi.org/10.1111/eth.12358
Méhu, M., & Dunbar, R. I. M. (2008). Relationship between smiling and laughter in humans (Homo sapiens): Testing the power asymmetry hypothesis. Folia Primatologica, 79(5), 269–280. https://doi.org/10.1159/000126928
Mehta, P. H., & Josephs, R. A. (2010). Testosterone and cortisol jointly regulate dominance: Evidence for a dual-hormone hypothesis. Hormones and Behavior, 58(5), 898–906. https://doi.org/10.1016/j.yhbeh.2010.08.020
Muñoz-Reyes, J. A., Polo, P., Valenzuela, N., Pavez, P., Ramírez-Herrera, O., Figueroa, O., Rodriguez-Sickert, C., Díaz, D., & Pita, M. (2020). The male warrior hypothesis: Testosterone-related cooperation and aggression in the context of intergroup conflict. Scientific Reports, 10(1), 375. https://doi.org/10.1038/s41598-019-57259-0
Neumann, C., Kulik, L., Agil, M., Engelhardt, A., & Widdig, A. (2022). Temporal dynamics and fitness consequences of coalition formation in male primates. Proceedings of the Royal Society B: Biological Sciences, 289(1976), 20212626. https://doi.org/10.1098/rspb.2021.2626
Phoenix, C. H., Goy, R. W., Gerall, A. A., & Young, W. C. (1959). Organizing action of prenatally administered testosterone propionate on the tissues mediating mating behavior in the female guinea pig. Endocrinology, 65(3), 369–382. https://doi.org/10.1210/endo-65-3-369
Preuschoft, S., & van Hooff, J. A. R. A. M. (1997). The social function of “smile” and “laughter”: Variations across primate species and societies. In U. Segerstråle & P. Molnár (Eds.), Nonverbal communication: Where nature meets culture (pp. 171–190). Lawrence Erlbaum.
Sherman, G. D., Rice, L. K., Jin, E. S., Jones, A. C., & Josephs, R. A. (2017). Sex differences in cortisol’s regulation of affiliative behavior. Hormones and Behavior, 92, 20–28. https://doi.org/10.1016/j.yhbeh.2016.12.005
Smith, J. E., Fichtel, C., Holmes, R. K., Kappeler, P. M., van Vugt, M., & Jaeggi, A. V. (2022). Sex bias in intergroup conflict and collective movements among social mammals: Male warriors and female guides. Philosophical Transactions of the Royal Society B: Biological Sciences, 377(1851), 20210142. https://doi.org/10.1098/rstb.2021.0142
Tamres, L. K., Janicki, D., & Helgeson, V. S. (2002). Sex differences in coping behavior: A meta-analytic review and an examination of relative coping. Personality and Social Psychology Review, 6(1), 2–30. https://doi.org/10.1207/S15327957PSPR0601_1
Thornton, J., Zehr, J. L., & Loose, M. D. (2009). Effects of prenatal androgens on rhesus monkeys: A model system to explore the organizational hypothesis in primates. Hormones and Behavior, 55(5), 633–645. https://doi.org/10.1016/j.yhbeh.2009.03.015
Waldman, B. (1987). Mechanisms of kin recognition. Journal of Theoretical Biology, 128(2), 159–185. https://doi.org/10.1016/S0022-5193(87)80167-4
Wallen, K. (2005). Hormonal influences on sexually differentiated behavior in nonhuman primates. Frontiers in Neuroendocrinology, 26(1), 7–26. https://doi.org/10.1016/j.yfrne.2005.02.001
Wrangham, R. W. (2018). Two types of aggression in human evolution. Proceedings of the National Academy of Sciences, 115(2), 245–253. https://doi.org/10.1073/pnas.1713611115