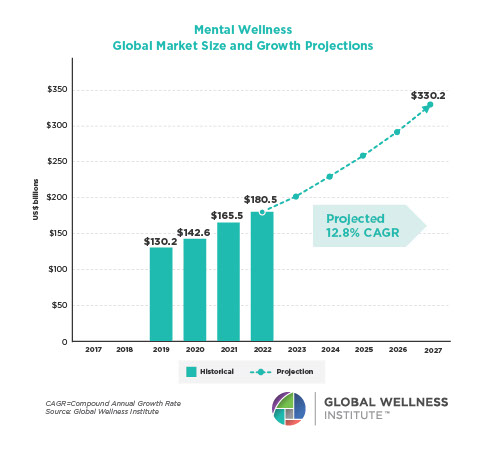
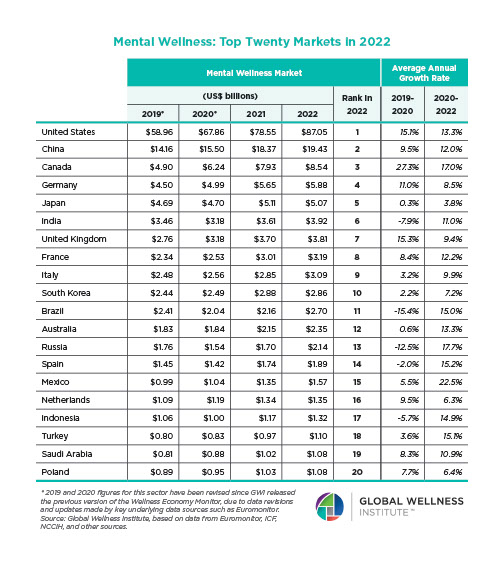
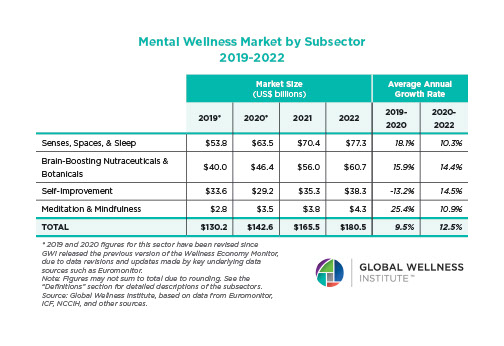
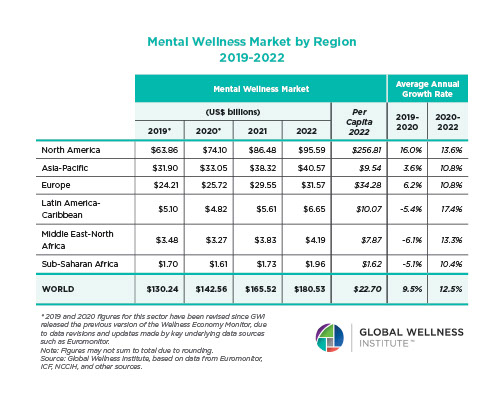
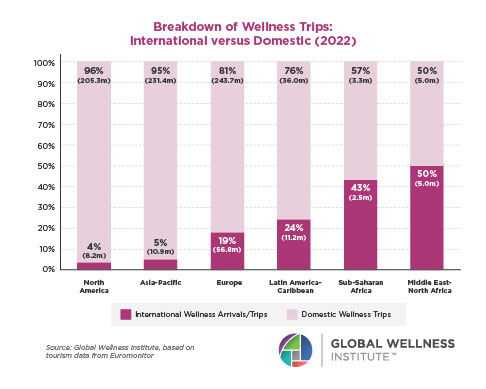
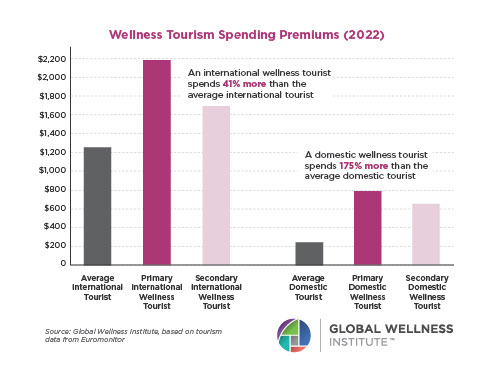
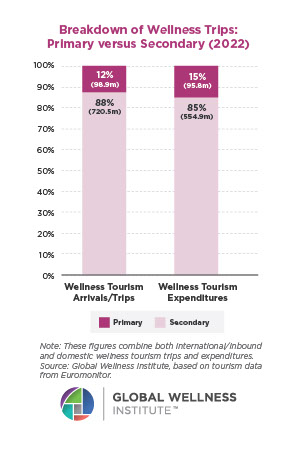
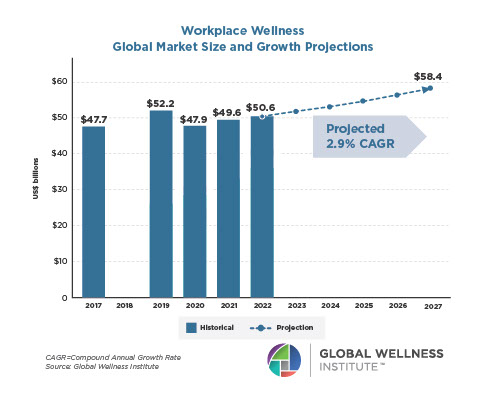
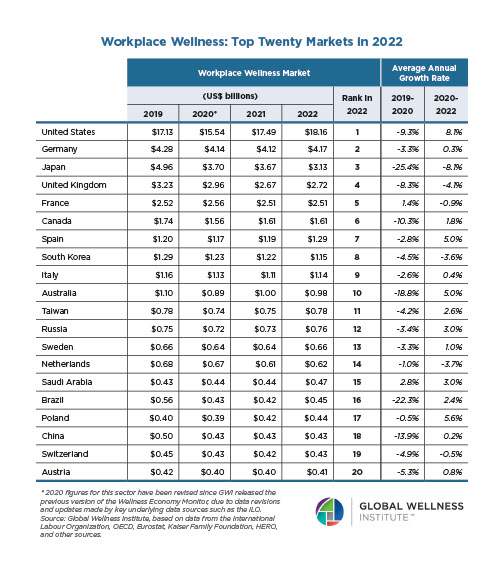
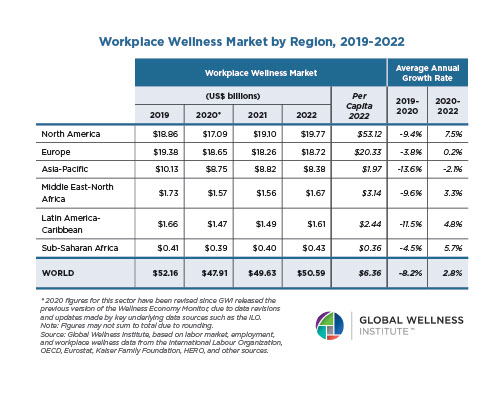
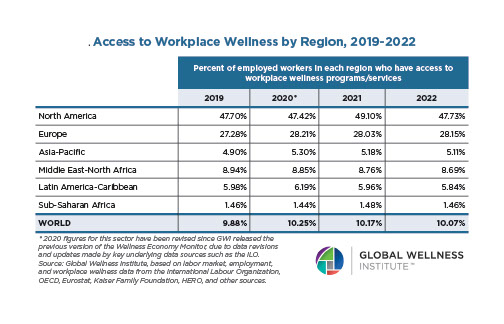
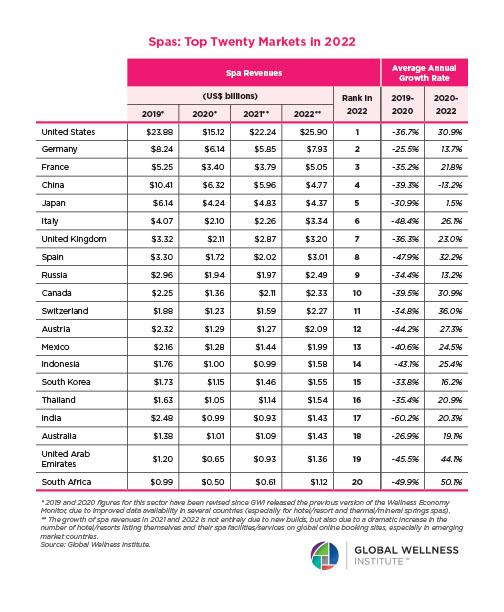
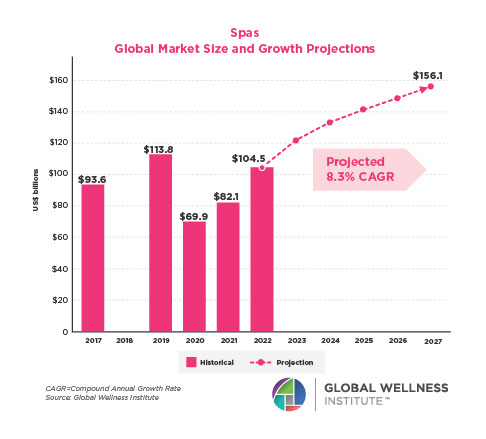
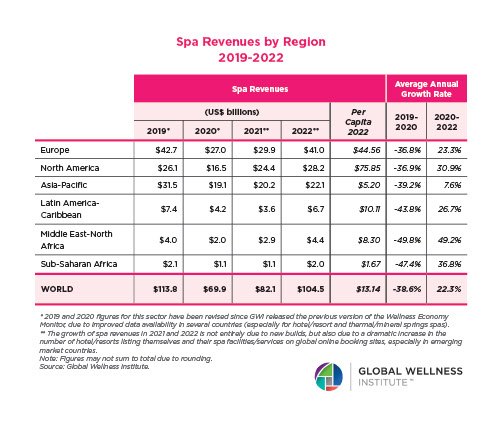
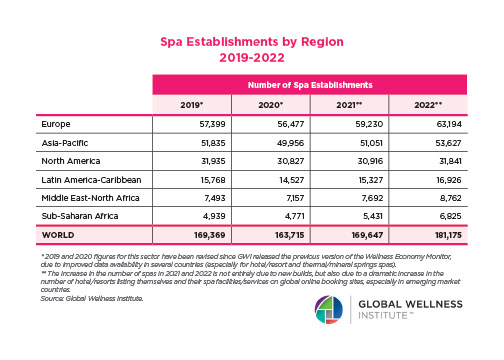
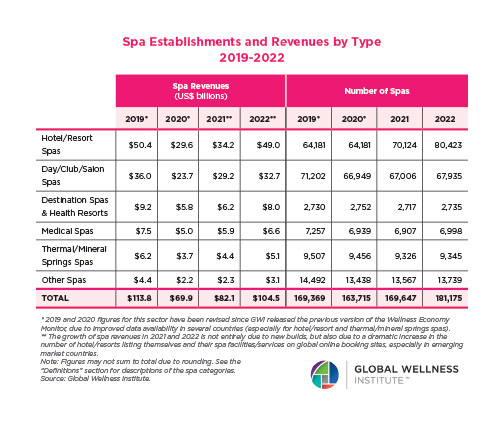
The Neural Architecture of Social Hierarchy and Male Group Behavior
Hierarchy as Cognitive Fodder
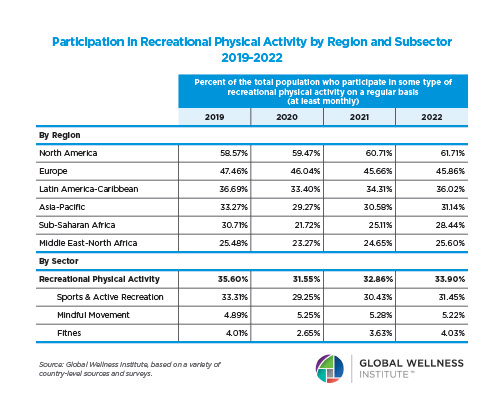
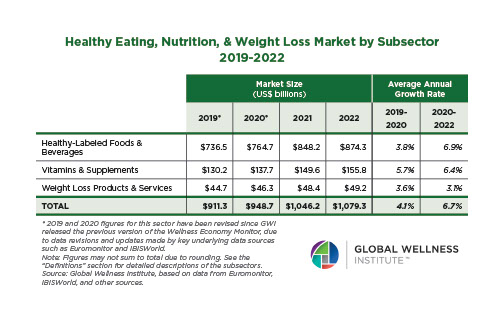
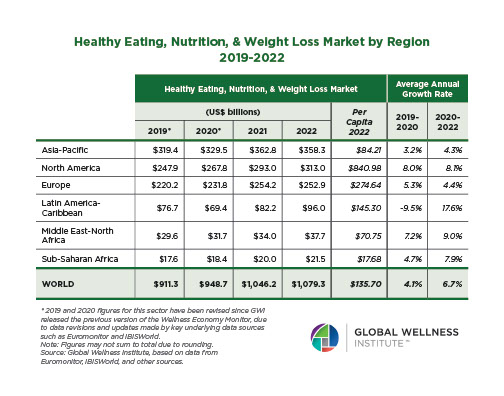
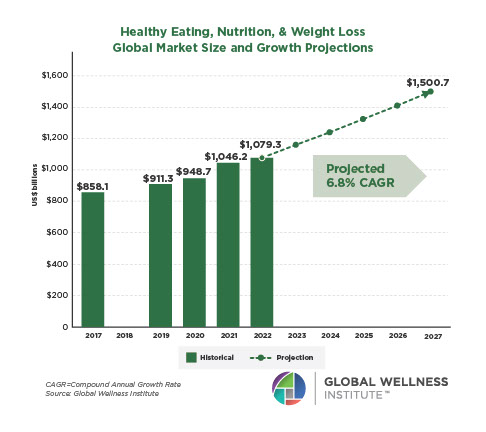
The behavioral repertoire described in the preceding essay — reactive and proactive aggression, affiliative approach and stress-withdrawal, cooperation and competition, dominance and subordination signaling, coalitional categorization — requires a continuously updated social map. Maintaining that map was one of the selection pressures that organized the primate brain. Across primates, ungulates, carnivores, cetaceans, and birds, species with larger and more complex social groups have disproportionately larger neocortices relative to body size (Dunbar, 1992, 1998; Shultz & Dunbar, 2007; Pérez-Barbería et al., 2007). The human neocortex — the outer, 2–4 mm thick layer of gray matter — is the largest relative to body size of any primate, reflecting an evolutionary history in which the ability to map social relationships, track reciprocity, infer rank, and predict the behavior of allies and rivals was under persistent selection.
Even human sleep architecture bears the imprint of group living. Humans sleep less than any other primate — roughly seven hours against a predicted 9.5 hours for a primate of our body mass — yet pack a higher proportion of REM sleep into that shorter period (Samson & Nunn, 2015). REM sleep is the phase most strongly implicated in memory consolidation, emotional regulation, and the processing of social information (Walker & Stickgold, 2006; Nunn & Samson, 2018). The selection pressures that compressed human sleep and enriched its REM proportion included predation risk from terrestrial sleeping, intergroup conflict, and the fitness benefits of longer waking hours for social interaction, skill transmission, and cultural learning (Samson & Nunn, 2015; Nunn et al., 2016). Sleep evolved not merely to restore the body, but to consolidate the cognitive demands of group cohabitation.
The Hierarchy-Tracking System
The cognitive demands of maintaining an accurate social map constitute a selection gradient along which the primate neocortex expanded. Functional neuroimaging has begun to identify the structures involved in hierarchy tracking and to show that they are computationally dissociable. The hippocampus and ventromedial prefrontal cortex (vmPFC) track the emergence of hierarchical knowledge in a domain-general fashion, coding linear rank structure regardless of whether the items being ranked are people or objects (Kumaran et al., 2012). The amygdala, however, is selectively recruited during social hierarchy learning, and individual differences in amygdala gray matter volume predict social transitive inference performance. The medial prefrontal cortex (mPFC) selectively mediates the updating of knowledge about one’s own position, as opposed to tracking the rank of others (Kumaran et al., 2016). Knowing the hierarchy requires relational memory and transitive inference; knowing your place in it requires integrating social feedback with self-referential processing.
The vmPFC sits at a convergence point between reward valuation, emotion regulation, and social cognition. Patients with focal vmPFC lesions exhibit impaired value-based decision-making despite intact general intelligence, diminished empathy and facial emotion recognition, and aberrant moral judgment (Hiser & Koenigs, 2018). Their social behavior deteriorates in specific ways: they approach angry faces rather than avoiding them, misjudge personal distance with strangers, and lose the capacity to update behavioral strategies based on social feedback (Rudebeck & Murray, 2014). When the damage occurs in childhood, the resulting antisocial behavior and moral impairment are more severe than when it occurs in adulthood, suggesting that the vmPFC organizes the developmental assembly of social computations during sensitive periods (Anderson et al., 2006). This developmental sensitivity will recur in the following essay on endocrine signaling, where prenatal and pubertal hormones act on these same regions during the windows when damage produces the most severe deficits.
The amygdala contributes a complementary function. Lesion patients show reduced attention to the eye region of faces and selective impairment in recognizing fearful expressions — a deficit that also characterizes children with psychopathic tendencies (Blair, 2008; Adolphs et al., 2005). The amygdala feeds reinforcement information forward to the vmPFC, which then represents expected outcomes to guide social decision-making. The vmPFC and amygdala thus form a bidirectional circuit in which threat and reward signals from the amygdala are integrated with value representations in the vmPFC to produce context-appropriate social behavior.
Behavioral variant frontotemporal dementia (bvFTD) offers a natural experiment in social-cognitive disassembly. The condition preferentially targets the anterior cingulate cortex (ACC) and frontoinsular cortex — the cortical hubs of what neuroscience now terms the salience network (Seeley, 2019). The cellular pathology begins with von Economo neurons, large projection neurons concentrated in these two regions, and spreads through the salience network’s connections to the amygdala, ventral striatum, and thalamus. Patients progressively lose empathy, social awareness, emotional responsiveness to others, and the capacity for moral reasoning, while other cognitive functions remain initially intact (Rascovsky et al., 2011). They become indifferent to the emotional states of family members, violate social norms without recognition that they have done so, and lose the motivation to initiate social interaction. The disease disassembles the social brain in the order its components were assembled, beginning with the phylogenetically most recent cortical salience hubs and progressing to the subcortical valuation structures that undergird social behavior.
The phylogenetic depth of the core hierarchy-tracking system extends well beyond primates. Male cichlid fish (Astatotilapia burtoni) can learn an implied dominance hierarchy among unfamiliar males by observation alone, performing transitive inference without direct interaction (Grosenick et al., 2007). This capacity in a teleost whose lineage diverged from mammals roughly 450 million years ago implies that hierarchy tracking is deeply conserved across vertebrate evolution. The cortical elaborations visible in primates — vmPFC self-referential integration, dorsolateral prefrontal cortex (DLPFC) strategic computation about when to challenge versus defer — are extensions built atop a subcortical valuation system (amygdala, ventral striatum, hypothalamic-pituitary axes) that is itself phylogenetically ancient.
Social experience selectively shapes these structures across the lifespan. A neuroimaging study of 23 macaques housed in social groups of different sizes found that living in larger groups produced increases in gray matter in the mid-superior temporal sulcus (mid-STS) and rostral prefrontal cortex, along with increased functional coupling between frontal and temporal cortex (Sallet et al., 2011). Gray matter differences in these regions also correlated with each animal’s dominance rank within its social network. A follow-up study by the same group identified dissociable neural circuits for social status: one circuit, centered on the amygdala, hypothalamus, and raphe nucleus region, showed greater gray matter in dominant individuals; another circuit mediated the relationship between dominance and social bond formation (Noonan et al., 2014). A subsequent study extended these findings to a free-ranging population of 68 rhesus macaques on Cayo Santiago, tracking five dimensions of social network structure against individual brain scans (Testard et al., 2022). The number of grooming partners — a direct measure of affiliative bonds — predicted gray matter volume in the mid-STS and ventral dysgranular insula, regions whose human homologues are implicated in social decision-making and empathy. Social status did not independently predict these specific structural differences once social network complexity was accounted for. The mid-STS region that increases with social network size in macaques has a connectional fingerprint partially similar to the human temporoparietal junction (TPJ), a region consistently implicated in theory of mind (Mars et al., 2012). Critically, scans of 35 juvenile and infant macaques in the Testard study showed no such brain-social correlations at birth; the structural differences emerged with development, suggesting that the social brain is shaped by social experience rather than being a fixed endowment. The mechanism by which social experience sculpts these regions is partly endocrine — affiliative interaction drives oxytocin release, which modulates receptor density in the regions the Testard study identified. The following essay on endocrine signaling specifies this pathway through the father-son biobehavioral synchrony evidence.
The question of whether these structures differ between male and female brains has received extensive meta-analytic attention. Ruigrok et al. (2014) conducted the first voxel-wise meta-analysis of sex differences in brain structure and found that males showed greater gray matter volume and density in the left amygdala, hippocampus, and insular cortex. A subsequent meta-analysis of amygdala volume across 58 MRI studies, however, found that the approximately 10% raw volume difference between males and females was fully explained by overall brain size; after correction for total intracranial volume, no significant sex difference remained (Marwha et al., 2017). A parallel meta-analysis reached the same conclusion for the hippocampus (Tan et al., 2016). The largest single-sample study to date, drawing on the UK Biobank, detected a modest amygdala effect (d = 0.18) after adjusting for total brain volume — statistically significant in a sample of that size, but small enough that the distributions between men and women overlap almost entirely (Ritchie et al., 2018). The sex differences pertinent to social behavior are functional rather than volumetric, driven through endocrine signaling.
The structures described in this section — amygdala, vmPFC, hypothalamus, ventral striatum, ACC — share a property beyond their social-cognitive functions: they are characterized by substantial expression of androgen receptors, oxytocin receptors, and glucocorticoid receptors (Simerly et al., 1990; Handa & Weiser, 2014). The distribution of these receptors is established during prenatal and pubertal development, creating a receptor landscape on which endocrine signals act across the lifespan. This receptor landscape is the interface between the neural circuitry described here and the hormonal signals described in the next essay.
Large-Scale Network Architecture
The individual structures described above are organized into three large-scale cortical networks whose interactions coordinate much of human social cognition. The default mode network (DMN) — anchored in medial prefrontal cortex, posterior cingulate cortex, precuneus, and angular gyrus — activates during self-referential processing, mentalizing about others, episodic memory retrieval, and the construction of internal narratives (Raichle, 2001; Greicius et al., 2003). The DMN constructs the mental representations of the social world — the internal model of who outranks whom, who is allied with whom, and where one stands relative to others. The salience network — anchored in the anterior insula and dorsal ACC, with subcortical extensions to the amygdala, hypothalamus, ventral striatum, and brainstem — detects stimuli, assigns them emotional weight, and coordinates the visceral and autonomic responses that accompany social evaluation (Seeley et al., 2007; Menon & Uddin, 2010). The frontoparietal control network (also called the executive control network) — anchored in the DLPFC and posterior parietal cortex — supports goal-directed behavior, cognitive control, and the strategic planning that determines whether an individual challenges, defers, cooperates, or withdraws (Seeley et al., 2007). The preceding essay described behavioral flexibility in which the same male cooperates with ingroup allies while aggressing against outgroup rivals depending on whether intergroup threat is salient. The triple-network model provides the computational framework for that context-dependent switching: the salience network functions as a switching mechanism between the other two, suppressing the internally directed DMN and engaging the externally directed executive system when a socially salient stimulus is detected (Menon & Uddin, 2010). When the salience network is selectively degraded, as in the bvFTD clinical picture described above, social cognition collapses even while executive function is initially preserved.
Comparative resting-state fMRI across primate species has begun to trace the evolutionary elaboration of these networks. Garin et al. (2022) applied comparable analytical methods to resting-state data from humans, macaques, marmosets, and mouse lemurs and identified large-scale functional networks in each species. The core network architecture was conserved across all four, but the DMN showed a marked structural gap between hominoid and non-hominoid primates: the medial prefrontal cortex and posterior cingulate cortex — tightly connected in the human DMN, forming the core axis that supports self-referential processing and internal mentation — were poorly connected in macaques, marmosets, and mouse lemurs. Instead, a fronto-temporal network involving the mPFC was detected consistently across non-hominoid species. The mPFC-PCC integration that characterizes the human DMN appears to be an evolutionary elaboration specific to the hominoid lineage, and its emergence coincides with the expansion of social-cognitive capacities that distinguish humans and great apes from other primates.
Neural Correlates of Behavior
The prior essay deconstructed male group behavior into five separable dimensions. Zink et al. (2008) identified the first major functional dissociation in how the human brain processes social rank, providing the neural context within which all five dimensions operate. In both stable and unstable hierarchies, the act of viewing a superior individual engaged the DLPFC and perceptual-attentional systems: the brain allocated more cognitive resources to monitoring individuals who could impose costs. In unstable hierarchies, additional regions were recruited: amygdala for threat evaluation, medial prefrontal cortex for social cognition, and motor-readiness areas for behavioral preparation. The ventral striatum activated for outcomes with hierarchical consequences, paralleling its role in reward processing. Social rank is processed through circuits that evolved for threat detection and reward valuation, elaborated with cortical oversight that permits strategic flexibility.
Reactive versus proactive aggression. Reactive aggression operates through amygdala-driven threat detection with impaired prefrontal regulation; serotonergic modulation dampens this pathway, and monoamine oxidase A (MAOA) genotype calibrates its sensitivity (Kolla & Bortolato, 2020). Proactive aggression is prefrontal-mediated and goal-directed, with low emotional arousal and sustained target focus (Wrangham, 2018). These forms of aggression can evolve independently because they run on distinct neural circuits. The pubertal window is when the MAOA × testosterone interaction becomes behaviorally consequential.
Affiliative approach versus stress-withdrawal. The prior essay characterized the male-typical stress response as affiliative withdrawal rather than undifferentiated fight-or-flight. Affiliative approach engages oxytocin-sensitive regions: the medial preoptic area, ventral striatum, and the vmPFC valuation circuit — the same regions that show increased activation in fathers viewing child stimuli, as the endocrine essay describes. Stress-withdrawal operates through a complementary pathway: glucocorticoid receptors in the amygdala amplify threat salience, while glucocorticoid action on the vmPFC suppresses the reward value of social approach. The salience network shifts processing toward threat monitoring rather than affiliative engagement. Elevated cortisol simultaneously suppresses testosterone-mediated competitive behavior through glucocorticoid antagonism of androgen receptor function and independently suppresses affiliative behavior in men, pushing the stressed male toward disengagement through parallel inhibition of both behavioral axes.
Cooperative versus competitive orientation. Cooperation between group members appears as inter-brain neural synchrony. Hyperscanning studies using functional near-infrared spectroscopy show that within-group neural synchronization in the DLPFC during competition predicts outgroup hostility, while synchronization in the TPJ during cooperation predicts outgroup likability (Lei et al., 2025; Zhang et al., 2023). Reduced DLPFC activity associates with stronger parochialism (Yang et al., 2020), suggesting that the prefrontal contribution to intergroup behavior is partially inhibitory — dampening ingroup favoritism and enforcing broader cooperative norms. The Muñoz-Reyes et al. (2020) finding described in the prior essay — that aggression and cooperation increase simultaneously under intergroup threat — is consistent with the dual-circuit hyperscanning evidence: DLPFC and TPJ synchronization can co-occur when the outgroup is salient, driving competitive and cooperative processing in parallel rather than trading off against each other.
Dominance and subordination signaling. Subordination signals — postural contraction, gaze aversion, vocal pitch elevation — were described in the prior essay as context-dependent communicative acts that maintain group membership while accepting lower rank. The neural pathway involves the anterior insula, which monitors the body’s status state through interoceptive processing, and the ACC, which detects the mismatch between expected and actual rank feedback (Zink et al., 2008). When the feedback signal mismatches expectations — when a male’s experienced status diverges from his internal model of his position — the ACC generates a conflict signal that drives behavioral adjustment. The resulting output, whether postural contraction or gaze aversion, is a computed behavioral response shaped by the mismatch, serving a function distinct from withdrawal: subordination signaling keeps the individual within the social field while reducing the probability of attack.
Coalitional categorization. The social categorization system recruits mPFC for self-group integration (Ma & Tan, 2023), fusiform face area for categorical processing of outgroup faces with reduced individuation (Amodio & Cikara, 2021), and TPJ for perspective-taking across the ingroup-outgroup boundary (Lei et al., 2025). The prior essay presented evidence that this system tracks alliance rather than phenotype — it evolved for coalitional partners determined by kinship and circumstance rather than chosen for compatibility. The neural regions involved in categorization and subordination signaling overlap substantially — mPFC, TPJ, anterior insula, and ACC participate in both — consistent with the preceding essay’s treatment of these as two subsystems of a single social-mapping capacity: a signaling channel that regulates rank and a categorization system that tracks coalitional affiliation.
These circuits function as partially dissociable modules whose relative activation depends on whether the context calls for competition, cooperation, affiliation, or withdrawal. The neural architecture implementing these behavioral dimensions is assembled through a sequence of hormonally organized developmental windows: prenatal testosterone establishes the receptor landscape on which pubertal testosterone acts; early caregiving calibrates the oxytocin system that governs the affiliative-withdrawal balance; and prefrontal maturation, not complete until the mid-twenties, gradually brings reactive aggression under cortical regulation. The following essay traces these developmental cascades and the endocrine signals that modulate the architecture in adulthood.
References
Adolphs, R., Gosselin, F., Buchanan, T. W., Tranel, D., Schyns, P., & Damasio, A. R. (2005). A mechanism for impaired fear recognition after amygdala damage. Nature, 433(7021), 68–72. https://doi.org/10.1038/nature03086
Amodio, D. M., & Cikara, M. (2021). The social neuroscience of prejudice. Annual Review of Psychology, 72, 439–469. https://doi.org/10.1146/annurev-psych-010419-050928
Anderson, S. W., Barrash, J., Bechara, A., & Tranel, D. (2006). Impairments of emotion and real-world complex behavior following childhood- or adult-onset damage to ventromedial prefrontal cortex. Journal of the International Neuropsychological Society, 12(2), 224–235. https://doi.org/10.1017/S1355617706060346
Blair, R. J. R. (2008). The amygdala and ventromedial prefrontal cortex: Functional contributions and dysfunction in psychopathy. Philosophical Transactions of the Royal Society B: Biological Sciences, 363(1503), 2557–2565. https://doi.org/10.1098/rstb.2008.0027
Dunbar, R. I. M. (1992). Neocortex size as a constraint on group size in primates. Journal of Human Evolution, 22(6), 469–493. https://doi.org/10.1016/0047-2484(92)90081-J
Dunbar, R. I. M. (1998). The social brain hypothesis. Evolutionary Anthropology, 6(5), 178–190. https://doi.org/10.1002/(SICI)1520-6505(1998)6:5%3C178::AID-EVAN5%3E3.0.CO;2-8
Garin, C. M., Hori, Y., Everling, S., Whitlow, C. T., Calabro, F. J., Luna, B., Froesel, M., Gacoin, M., Ben Hamed, S., Dhenain, M., & Constantinidis, C. (2022). An evolutionary gap in primate default mode network organization. Cell Reports, 39(2), 110669. https://doi.org/10.1016/j.celrep.2022.110669
Greicius, M. D., Krasnow, B., Reiss, A. L., & Menon, V. (2003). Functional connectivity in the resting brain: A network analysis of the default mode hypothesis. Proceedings of the National Academy of Sciences, 100(1), 253–258. https://doi.org/10.1073/pnas.0135058100
Grosenick, L., Clement, T. S., & Fernald, R. D. (2007). Fish can infer social rank by observation alone. Nature, 445(7126), 429–432. https://doi.org/10.1038/nature05511
Handa, R. J., & Weiser, M. J. (2014). Gonadal steroid hormones and the hypothalamo-pituitary-adrenal axis. Frontiers in Neuroendocrinology, 35(2), 197–220. https://doi.org/10.1016/j.yfrne.2013.11.001
Hiser, J., & Koenigs, M. (2018). The multifaceted role of the ventromedial prefrontal cortex in emotion, decision making, social cognition, and psychopathology. Biological Psychiatry, 83(8), 638–647. https://doi.org/10.1016/j.biopsych.2017.10.030
Kolla, N. J., & Bortolato, M. (2020). The role of monoamine oxidase A in the neurobiology of aggressive, antisocial, and violent behavior: A tale of mice and men. Progress in Neurobiology, 194, 101875. https://doi.org/10.1016/j.pneurobio.2020.101875
Kumaran, D., Melo, H. L., & Duzel, E. (2012). The emergence and representation of knowledge about social and nonsocial hierarchies. Neuron, 76(3), 653–666. https://doi.org/10.1016/j.neuron.2012.09.035
Kumaran, D., Banino, A., Blundell, C., Hassabis, D., & Dayan, P. (2016). Computations underlying social hierarchy learning: Distinct neural mechanisms for updating and representing self-relevant information. Neuron, 92(5), 1135–1147. https://doi.org/10.1016/j.neuron.2016.10.052
Lei, Y., Liu, S., Guo, X., & Zuo, B. (2025). Neural synchronization and its impact on intergroup attitudes in dynamic interactions. Communications Biology, 8(1), 314. https://doi.org/10.1038/s42003-025-07776-8
Ma, Y., & Tan, H. (2023). Representation of intergroup conflict in the human brain. Neuron, 111(11), 1692–1696. https://doi.org/10.1016/j.neuron.2023.05.004
Mars, R. B., Neubert, F.-X., Noonan, M. P., Sallet, J., Toni, I., & Rushworth, M. F. S. (2012). On the relationship between the “default mode network” and the “social brain”. Frontiers in Human Neuroscience, 6, 189. https://doi.org/10.3389/fnhum.2012.00189
Marwha, D., Halari, M., & Eliot, L. (2017). Meta-analysis reveals a lack of sexual dimorphism in human amygdala volume. NeuroImage, 147, 282–294. https://doi.org/10.1016/j.neuroimage.2016.12.021
Menon, V., & Uddin, L. Q. (2010). Saliency, switching, attention and control: A network model of insula function. Brain Structure and Function, 214(5–6), 655–667. https://doi.org/10.1007/s00429-010-0262-0
Muñoz-Reyes, J. A., Polo, P., Valenzuela, N., Pavez, P., Ramírez-Herrera, O., Figueroa, O., Rodriguez-Sickert, C., Díaz, D., & Pita, M. (2020). The male warrior hypothesis: Testosterone-related cooperation and aggression in the context of intergroup conflict. Scientific Reports, 10(1), 375. https://doi.org/10.1038/s41598-019-57259-0
Noonan, M. P., Sallet, J., Mars, R. B., Neubert, F. X., O’Reilly, J. X., Andersson, J. L., Mitchell, A. S., Bell, A. H., Miller, K. L., & Rushworth, M. F. S. (2014). A neural circuit covarying with social hierarchy in macaques. PLOS Biology, 12(9), e1001940. https://doi.org/10.1371/journal.pbio.1001940
Nunn, C. L., & Samson, D. R. (2018). Sleep in a comparative context: Investigating how human sleep differs from sleep in other primates. American Journal of Physical Anthropology, 166(3), 601–612. https://doi.org/10.1002/ajpa.23427
Nunn, C. L., Samson, D. R., & Krystal, A. D. (2016). Shining evolutionary light on human sleep and sleep disorders. Evolution, Medicine, and Public Health, 2016(1), 227–243. https://doi.org/10.1093/emph/eow018
Pérez-Barbería, F. J., Shultz, S., & Dunbar, R. I. M. (2007). Evidence for coevolution of sociality and relative brain size in three orders of mammals. Evolution, 61(12), 2811–2821. https://doi.org/10.1111/j.1558-5646.2007.00229.x
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001). A default mode of brain function. Proceedings of the National Academy of Sciences, 98(2), 676–682. https://doi.org/10.1073/pnas.98.2.676
Rascovsky, K., Hodges, J. R., Knopman, D., Mendez, M. F., Kramer, J. H., Neuhaus, J., van Swieten, J. C., Seelaar, H., Dopper, E. G. P., Onyike, C. U., Hillis, A. E., Josephs, K. A., Boeve, B. F., Kertesz, A., Seeley, W. W., Rankin, K. P., Johnson, J. K., Gorno-Tempini, M.-L., Rosen, H., … Miller, B. L. (2011). Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain, 134(9), 2456–2477. https://doi.org/10.1093/brain/awr179
Ritchie, S. J., Cox, S. R., Shen, X., Lombardo, M. V., Reus, L. M., Alloza, C., Harris, M. A., Alderson, H. L., Hunter, S., Neilson, E., Liewald, D. C. M., Auyeung, B., Whalley, H. C., Lawrie, S. M., Gale, C. R., Bastin, M. E., McIntosh, A. M., & Deary, I. J. (2018). Sex differences in the adult human brain: Evidence from 5,216 UK Biobank participants. Cerebral Cortex, 28(8), 2959–2975. https://doi.org/10.1093/cercor/bhy109
Rudebeck, P. H., & Murray, E. A. (2014). The orbitofrontal oracle: Cortical mechanisms for the prediction and evaluation of specific behavioral outcomes. Neuron, 84(6), 1143–1156. https://doi.org/10.1016/j.neuron.2014.10.049
Ruigrok, A. N. V., Salimi-Khorshidi, G., Lai, M.-C., Baron-Cohen, S., Lombardo, M. V., Tait, R. J., & Suckling, J. (2014). A meta-analysis of sex differences in human brain structure. Neuroscience & Biobehavioral Reviews, 39, 34–50. https://doi.org/10.1016/j.neubiorev.2013.12.004
Sallet, J., Mars, R. B., Noonan, M. P., Andersson, J. L., O’Reilly, J. X., Jbabdi, S., Croxson, P. L., Jenkinson, M., Miller, K. L., & Rushworth, M. F. S. (2011). Social network size affects neural circuits in macaques. Science, 334(6056), 697–700. https://doi.org/10.1126/science.1210027
Samson, D. R., & Nunn, C. L. (2015). Sleep intensity and the evolution of human cognition. Evolutionary Anthropology, 24(6), 225–237. https://doi.org/10.1002/evan.21464
Seeley, W. W. (2019). The salience network: A neural system for perceiving and responding to homeostatic demands. Journal of Neuroscience, 39(50), 9878–9882. https://doi.org/10.1523/JNEUROSCI.1138-17.2019
Seeley, W. W., Menon, V., Schatzberg, A. F., Keller, J., Glover, G. H., Kenna, H., Reiss, A. L., & Greicius, M. D. (2007). Dissociable intrinsic connectivity networks for salience processing and executive control. Journal of Neuroscience, 27(9), 2349–2356. https://doi.org/10.1523/JNEUROSCI.5587-06.2007
Shultz, S., & Dunbar, R. I. M. (2007). The evolution of the social brain: Anthropoid primates contrast with other vertebrates. Proceedings of the Royal Society B, 274(1624), 2429–2436. https://doi.org/10.1098/rspb.2007.0693
Simerly, R. B., Chang, C., Muramatsu, M., & Swanson, L. W. (1990). Distribution of androgen and estrogen receptor mRNA-containing cells in the rat brain: An in situ hybridization study. Journal of Comparative Neurology, 294(1), 76–95. https://doi.org/10.1002/cne.902940107
Tan, A., Ma, W., Vira, A., Marwha, D., & Eliot, L. (2016). The human hippocampus is not sexually-dimorphic: Meta-analysis of structural MRI volumes. NeuroImage, 124 (Pt A), 350–366. https://doi.org/10.1016/j.neuroimage.2015.08.050
Testard, C., Brent, L. J. N., Andersson, J., Chiou, K. L., Negron-Del Valle, J. E., DeCasien, A. R., Acevedo-Ithier, A., Stock, M. K., Antón, S. C., Gonzalez, O., Walker, C. S., Foxley, S., Compo, N. R., Bauman, S., Ruiz-Lambides, A. V., Martinez, M. I., Skene, J. H. P., Horvath, J. E., Cayo Biobank Research Unit, … Sallet, J. (2022). Social connections predict brain structure in a multidimensional free-ranging primate society. Science Advances, 8(15), eabl5794. https://doi.org/10.1126/sciadv.abl5794
Walker, M. P., & Stickgold, R. (2006). Sleep, memory, and plasticity. Annual Review of Psychology, 57, 139–166. https://doi.org/10.1146/annurev.psych.56.091103.070307
Wrangham, R. W. (2018). Two types of aggression in human evolution. Proceedings of the National Academy of Sciences, 115(2), 245–253. https://doi.org/10.1073/pnas.1713611115
Yang, J., Zhang, H., Ni, J., De Dreu, C. K. W., & Ma, Y. (2020). Within-group synchronization in the prefrontal cortex associates with intergroup conflict. Nature Neuroscience, 23(6), 754–760. https://doi.org/10.1038/s41593-020-0630-x
Zhang, H., Yang, J., Ni, J., De Dreu, C. K. W., & Ma, Y. (2023). Leader–follower behavioural coordination and neural synchronization during intergroup conflict. Nature Human Behaviour, 7(11), 1972–1981. https://doi.org/10.1038/s41562-023-01663-0
Zink, C. F., Tong, Y., Chen, Q., Bassett, D. S., Stein, J. L., & Meyer-Lindenberg, A. (2008). Know your place: Neural processing of social hierarchy in humans. Neuron, 58(2), 273–283. https://doi.org/10.1016/j.neuron.2008.01.025