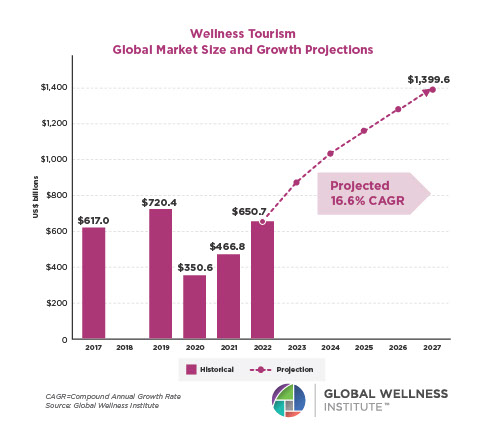
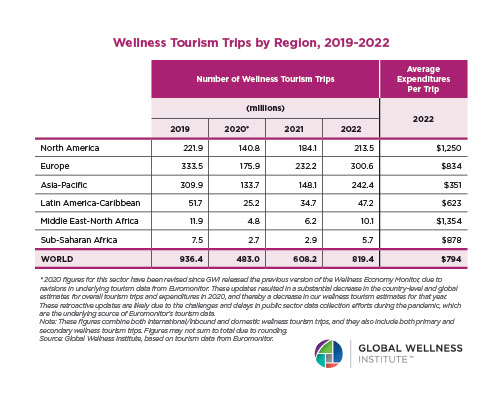
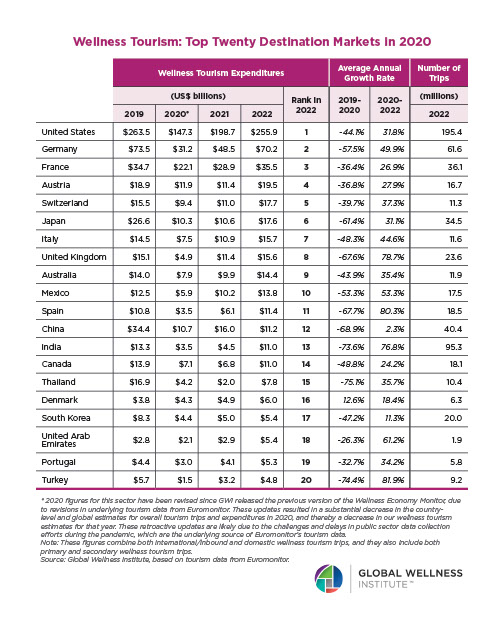
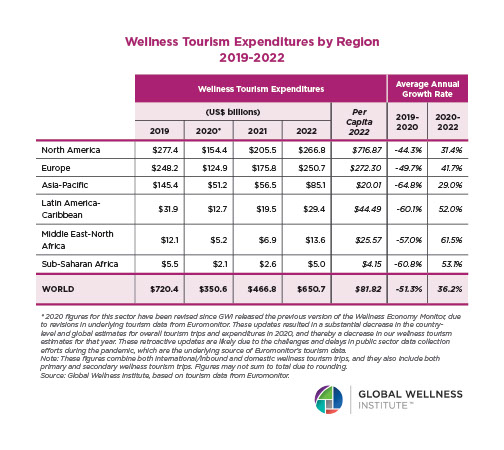
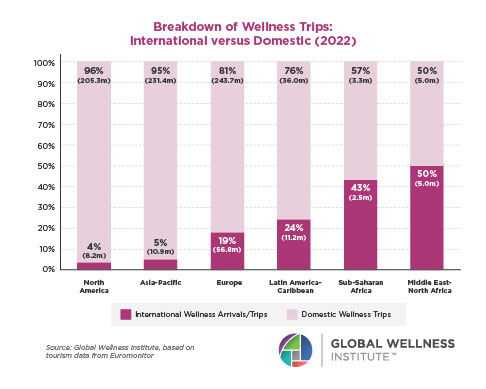
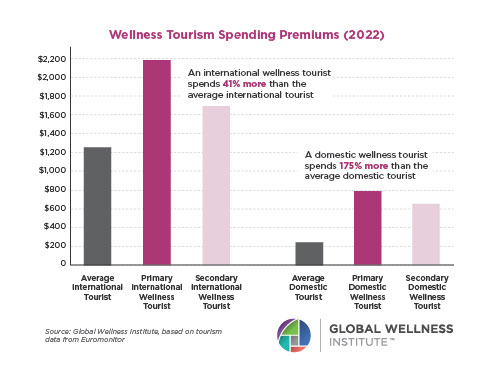
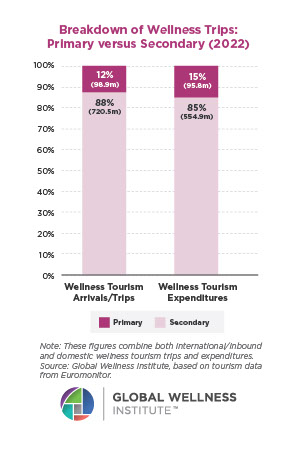
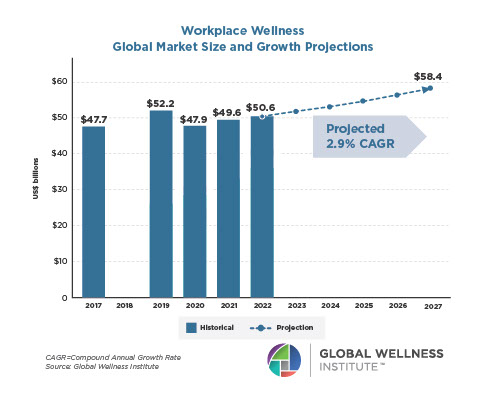
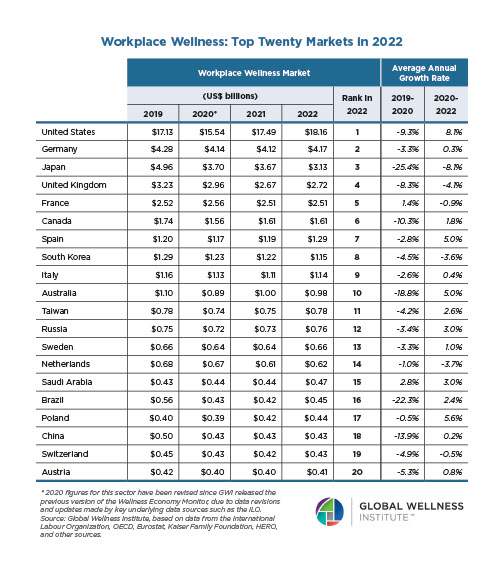
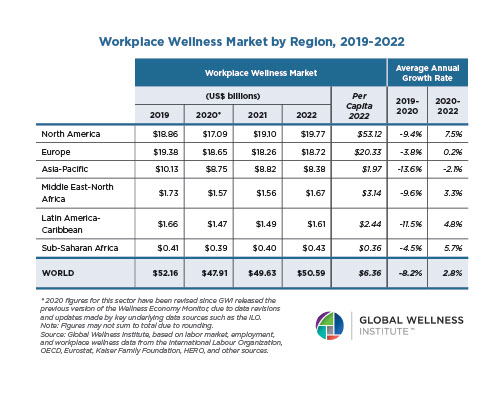
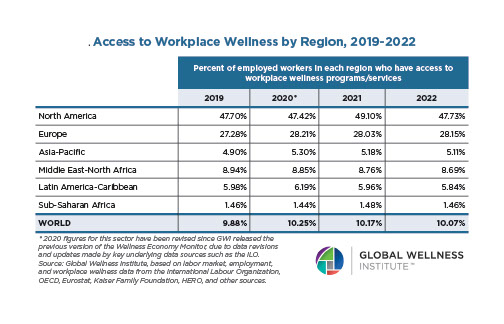
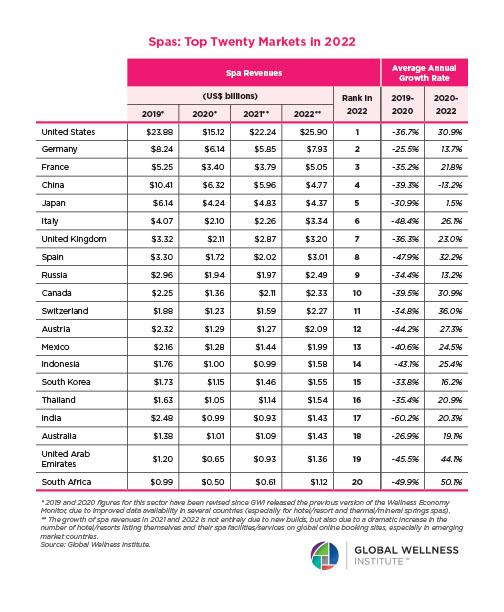
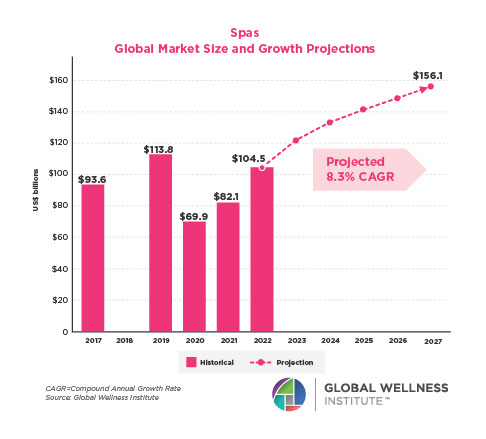
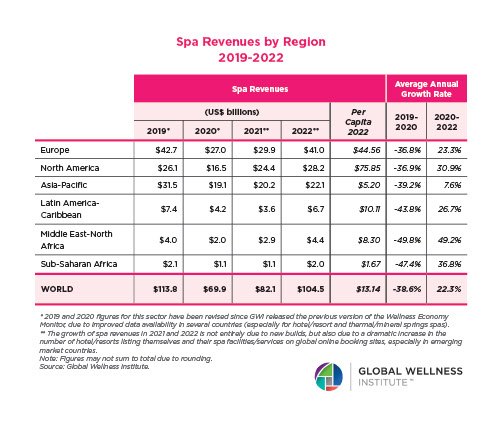
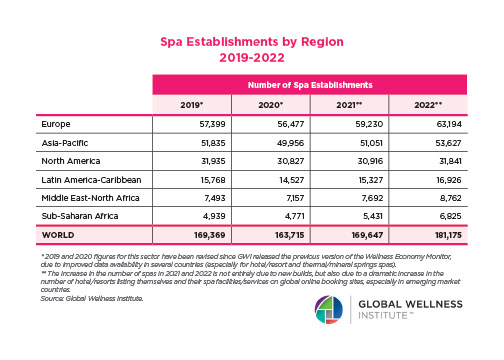
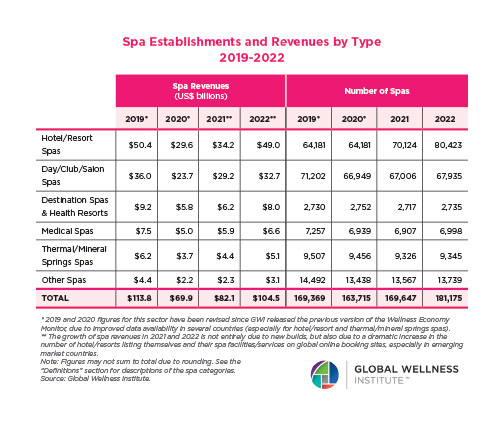
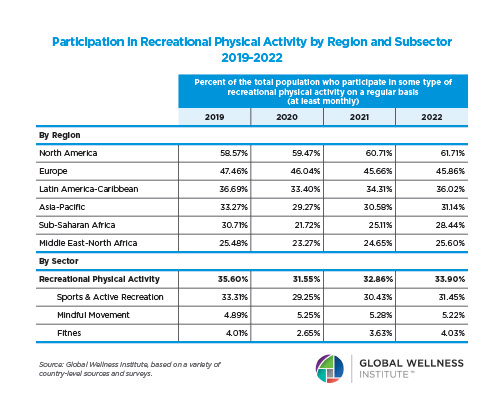
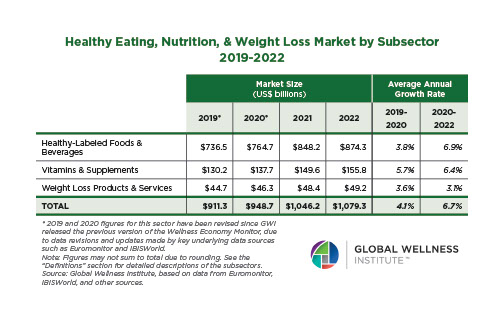
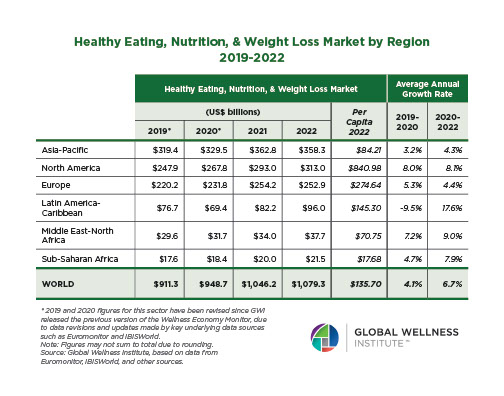
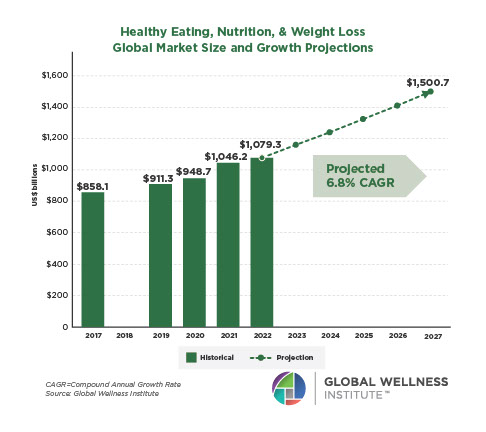
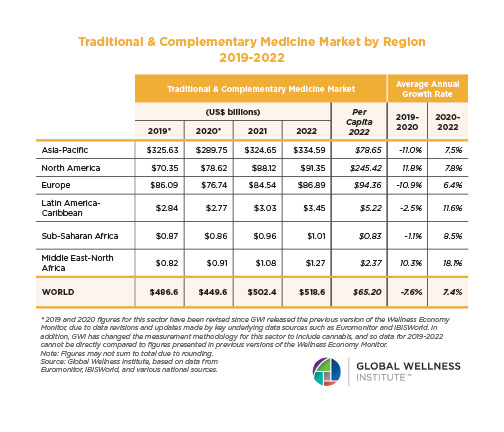
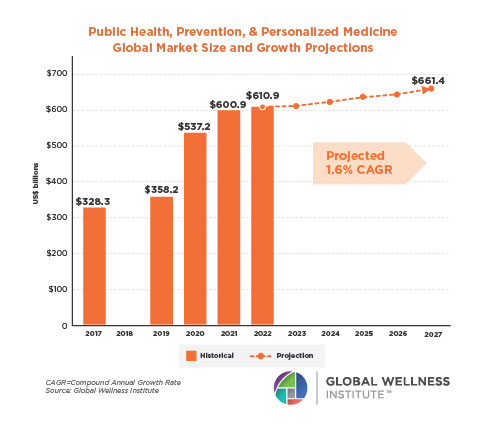
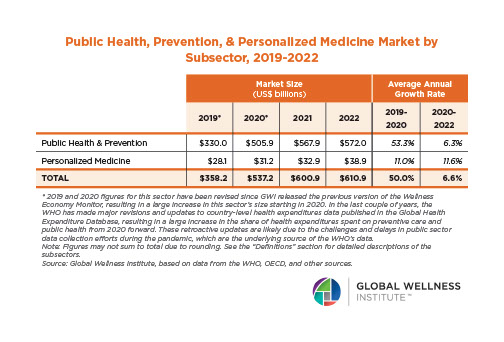
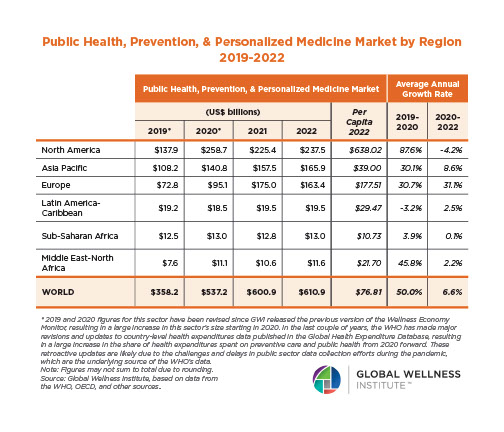
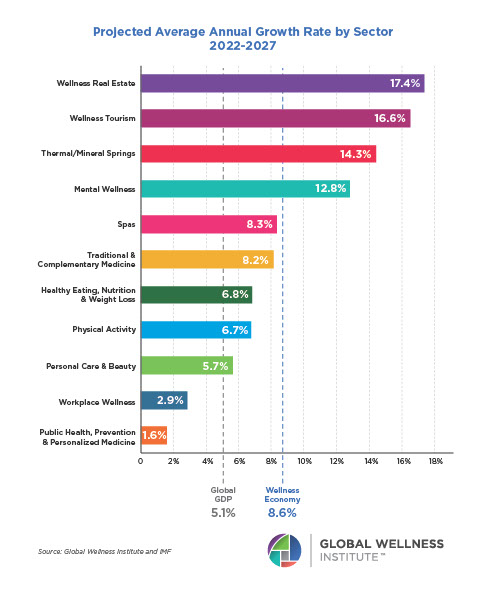
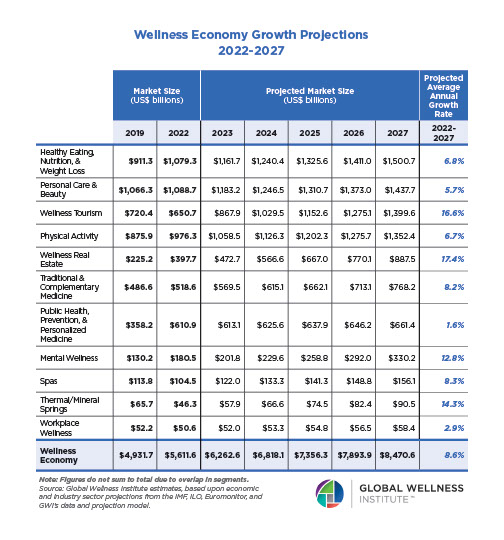
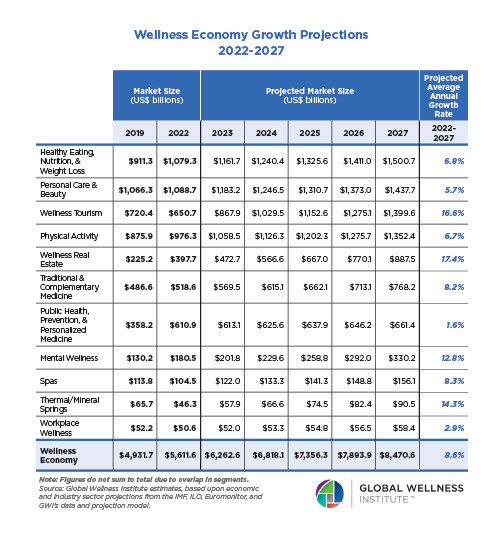
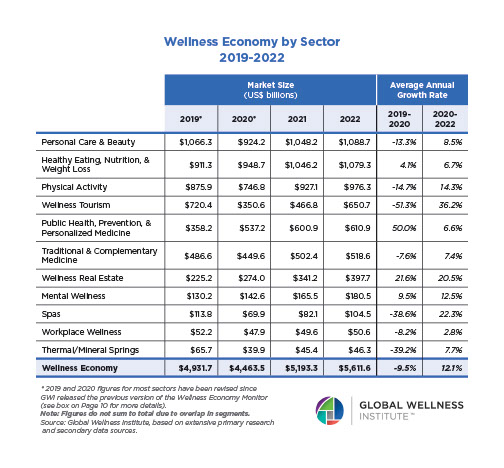
Endocrine Signaling Through the Male Social Brain
The neural architecture of male social behavior — the hierarchy-tracking, categorization, and behavioral-selection circuits described in the preceding essay — is calibrated and recalibrated by endocrine signals across the lifespan. The structures that compose that architecture — amygdala, vmPFC, hypothalamus, ventral striatum, ACC — share a property beyond their social-cognitive functions: they express androgen receptors, oxytocin receptors, and glucocorticoid receptors at substantial density (Simerly et al., 1990; Handa & Weiser, 2014). This receptor landscape is the interface through which endocrine signals organize the circuits during development and modulate them in adulthood. The organizing framework is a behavioral space (Figure 4) defined by two axes — testosterone-mediated competitive processing and oxytocin-mediated affiliative processing — with cortisol gating expression along both. A man’s position in this space shifts with developmental stage, social context, and ecological conditions. The sections that follow trace how the initial coordinates get set and how they change.

Developmental Windows: How Endocrine Timing Patterns the Architecture
The same hormones that modulate adult social behavior also organize the receptor distribution and circuit connectivity they will later modulate, through a sequence of developmental windows with distinct endocrine profiles and distinct neural targets.
Prenatal testosterone, produced by the fetal testes following SRY activation during the first and second trimesters, masculinizes the brain’s organizational structure. In humans, this operates primarily through direct androgen receptor action rather than the aromatization-to-estradiol pathway that predominates in rodents (Wilson & Davies, 2007). The targets include amygdala volume, hypothalamic nuclei involved in reproductive and aggressive behavior, and the density and distribution of androgen and estrogen receptors across cortical and subcortical regions (Auyeung et al., 2013; Hines, 2020). This prenatal exposure creates the receptor landscape — the physical distribution of androgen receptors, oxytocin receptors, and glucocorticoid receptors across brain regions — on which all subsequent hormonal signals will act. A postnatal testosterone surge during the first six months of life (“mini-puberty”) further consolidates these organizational effects, though its specific behavioral consequences remain poorly characterized in humans.
Puberty represents the second major organizational window. The pubertal testosterone surge activates the scaffolding laid down prenatally: it drives facial masculinization, muscularity, and voice-pitch lowering, and at the neural level it blunts HPA axis reactivity (Plant & Barker-Gibb, 2004) and recalibrates dopaminergic homeostasis in the midbrain (Kolla & Bortolato, 2020). This recalibration has consequences for aggression sensitivity, because the dopamine system modulates both reward processing and impulsivity. The pubertal window is also when the interaction between testosterone and MAOA genotype becomes behaviorally consequential: MAOA-deficient individuals show heightened sensitivity to the dopaminergic effects of pubertal testosterone, potentially amplifying reactive aggression in the presence of environmental adversity (Kolla & Bortolato, 2020).
The developmental windows illustrate that the same hormonal signal produces different behavioral outcomes depending on the receptor sensitivity of the target tissue, which is set by genetic variation. Two loci introduced in the Y chromosome essay exemplify this. The androgen receptor (AR) CAG repeat modulates cellular sensitivity to testosterone — shorter repeats produce greater transcriptional activity — with behavioral consequences that are population-specific: among Hadza foragers and Datoga pastoralists, shorter repeats predicted greater aggression and mediated the relationship between repeat length and reproductive output (Butovskaya et al., 2015), while in Western populations the same association is frequently null (Valenzuela et al., 2022). The inconsistency is consistent with gene × environment interaction in which the ecology — whether status is achieved through physical competition or through prestige — determines whether the genotype’s behavioral expression is amplified or silent. MAOA operates through an analogous logic: the low-activity variant lowers the threshold for reactive aggression, but only in individuals who experienced childhood maltreatment (Caspi et al., 2002; Byrd & Manuck, 2014, meta-analysis), and its interaction with pubertal testosterone amplifies the effect through dopaminergic dysregulation. The result in both cases is a three-way interaction — gene × endocrine × environment — in which the genetic variant sets the receptor sensitivity, the endocrine signal activates the system, and the environmental input determines whether the activation exceeds the threshold for behavioral expression.
The Father’s Endocrine Transition and Its Effect on the Developing Son
Fatherhood initiates a distinct endocrine reorganization in men: testosterone declines and oxytocin rises, with the magnitude of both changes proportional to the degree of paternal involvement (Gettler et al., 2011; Mascaro et al., 2014). This shift moves the father upward and leftward in the behavioral space — from competitive-dominant territory toward caregiver-oriented territory — and it is accompanied by neural changes: fathers show stronger activation in mentalizing regions (TPJ), face-emotion processing areas, and reward circuitry (medial OFC) when viewing child stimuli (Mascaro et al., 2014).
The effect on the developing son arrives through biobehavioral synchrony. Feldman et al. (2010) demonstrated that after synchronous father-infant interaction — temporally coordinated gaze, touch, and vocalization — both father and infant showed coordinated oxytocin release. Weisman et al. (2012) showed that administering intranasal oxytocin to fathers increased not only paternal oxytocin and caregiving behavior but also the infant’s oxytocin levels and social engagement. The father-child dyad functions as a neurochemical feedback loop in which the father’s endocrine state directly calibrates the son’s developing oxytocin system.
Gleason and Marler (2013) showed in biparental California mice that pups raised by high-caregiving fathers became high-caregiving parents themselves, even when cross-fostered to different biological parents. The effect was traceable to oxytocin receptor density in the medial preoptic area — a region critical for parental behavior. Sons of involved fathers developed denser oxytocin receptor fields in this region, which in turn set the baseline sensitivity of the affiliative system for life. The father’s postpartum endocrine transition increases oxytocin receptor density in the son’s medial preoptic area, shifting the son’s default position in the behavioral space toward higher affiliative processing. When the father’s endocrine transition fails to occur — when testosterone does not decline and oxytocin does not rise, as may happen with absent or disengaged fathers — the son’s oxytocin system is calibrated to a lower baseline, producing a default position shifted toward the bottom half of the behavioral space (lower affiliation, higher relative competitive processing or withdrawal).
Testosterone and Oxytocin
With the receptor distribution established, the question becomes how testosterone and oxytocin modulate the system in adulthood. Crespi (2016) assembled the most comprehensive formal model, treating the two hormones as opposing vectors of social cognition. In this model, testosterone administration reduces orbitofrontal cortex connectivity with the amygdala — connectivity that would enhance social-emotional input into decision-making — while oxytocin administration increases it. The model predicts that testosterone would reduce performance on tasks requiring mentalizing, such as reading emotions from the eye region of faces, whereas oxytocin would improve performance. Two subsequent large-scale double-blind RCTs in men (n = 243 and n = 400), however, found no main effect of testosterone on Reading the Mind in the Eyes Test performance (Nadler et al., 2019).
The evidence on testosterone and prosocial behavior elucidates the model’s inconsistency. Testosterone administration reduces trust toward unfamiliar others, while oxytocin administration increases it (Bos et al., 2010; Kosfeld et al., 2005). The effect is robust across multiple paradigms and has been independently replicated. Concerning prosocial behavior, 192 men given testosterone or placebo and measured on prosocial learning under observed and private conditions showed a specific pattern: testosterone eliminated the audience effect — the tendency to behave more generously when watched — without degrading prosocial learning itself (Kutlikova et al., 2023). Computational modeling traced the mechanism to reduced choice consistency for prosocial options under observation, and the effect was more pronounced in men higher in trait dominance. Testosterone, on this evidence, suppresses the performance of generosity for reputational gain rather than diminishing affiliative motivation per se. Exogenous testosterone also reduces generosity toward socially distant others through reduced temporoparietal junction (TPJ) activity, the region that supports perspective-taking and other-oriented computation (Ou et al., 2021). The opposition with oxytocin in this domain operates through status computation and self-other weighting rather than through a simple suppression of prosociality. Overall, the literature on testosterone and social interaction treats empathy reduction as one component of a status-seeking motivational shift rather than as a direct cognitive impairment (Eisenegger et al., 2011; Bos et al., 2012). Testosterone alters when and toward whom social cognition is deployed, with status-relevant contexts showing the clearest effects.
Oxytocin’s behavioral effects are similarly bounded. Intranasal oxytocin administration increased cooperation toward ingroup members and simultaneously increased defensive aggression toward outgroup threats in an intergroup resource-competition paradigm; the effect was parochial prosociality, favoring one’s own group at the expense of the other, rather than generalized warmth (De Dreu et al., 2010). A subsequent study extended this to moral cognition, with oxytocin biasing male participants’ judgments in sacrificial dilemmas toward sparing ingroup over outgroup targets (De Dreu et al., 2011). Testosterone runs in parallel: among fifty male soccer fans playing a single-shot ultimatum game against fans of their own team and fans of rival teams under neutral and competitive framing, endogenous testosterone predicted both greater generosity toward ingroup proposers and higher rejection rates of outgroup offers — including rejection of fair 40%-of-endowment offers — when the context was competitive (Diekhof et al., 2014). Both hormones promote parochial rather than generalized prosociality, through distinct mechanisms: testosterone triggers status computation, while oxytocin triggers bonding.
The neural targets of the two hormones reflect this functional distinction. Oxytocin acts primarily through receptors in the hypothalamus, nucleus accumbens, ventral tegmental area, and amygdala — the reward and salience circuitry that underlies bonding, social recognition, and ingroup preference (De Dreu et al., 2010, 2016). Testosterone acts through androgen receptors distributed across the amygdala, lateral septum, orbitofrontal cortex (OFC), dorsolateral prefrontal cortex, and periaqueductal gray — regions involved in threat evaluation, status computation, and behavioral readiness. The two hormones converge on the amygdala with opposing functional consequences. Exogenous testosterone reduces connectivity between the amygdala and the OFC, decoupling social-emotional input from prefrontal regulation (van Wingen et al., 2010). Oxytocin increases this connectivity, strengthening top-down modulation of amygdala output toward affiliative interpretation (Kirsch et al., 2005). Terburg and van Honk (2013) proposed a multilevel account of the testosterone side: the hormone inhibits acute fear responses at the basolateral amygdala, promotes reactive dominance through vasopressin gene-expression changes in the central-medial amygdala, and reduces cortical control over amygdala output — three mechanisms operating at different timescales, all oriented toward status acquisition and maintenance. This convergence on the amygdala with divergent functional consequences is the neural basis of the two-axis structure in the behavioral space figure. Since testosterone suppresses strategic prosociality — reputation-managed generosity — rather than affiliative capacity, a man can occupy the high-testosterone, high-oxytocin quadrant (parochial altruist) without contradiction: in this regime, his affiliation is intrinsically rather than performatively motivated.
Cortisol as Gating Signal
Cortisol does not define a separate behavioral axis so much as it gates the expression of the other two. Mehta and Josephs (2010) formalized this as the dual-hormone hypothesis: testosterone predicts dominant, status-seeking behavior only when cortisol is low. When cortisol is elevated, testosterone’s behavioral effects are attenuated, and the individual shifts toward avoidance rather than competitive engagement. The neural mechanism involves glucocorticoid receptor action in the hippocampus, prefrontal cortex, and amygdala, where cortisol suppresses hypothalamic-pituitary-gonadal (HPG) axis activity, antagonizes androgen receptor function at the tissue level, and downregulates androgen receptor expression (Viau, 2002; Burnstein et al., 1995).
The meta-analytic evidence on the dual-hormone hypothesis is supportive in direction but modest in magnitude. Dekkers et al. (2019) synthesized 33 studies (n = 8,538) and found a significant but small interaction effect of testosterone and cortisol on status-relevant behavior (r = −.061), with the strongest effects appearing for direct measures of social status and in male samples. The authors identified signs of publication bias and analytic flexibility across the constituent studies, and the originators of the hypothesis themselves describe the cumulative evidence as promising but inconsistent, calling for large preregistered replications (Knight et al., 2020).
Cortisol’s gating function extends beyond the competitive channel. Sherman et al. (2017) measured cortisol change and affiliative behavior in men and women following a competitive event and found a striking sex difference. Men were more affiliative after victory; women were more affiliative after defeat. The cortisol-affiliation relationship was itself sex-differentiated: in women, greater cortisol increase predicted greater affiliation, consistent with a tend-and-befriend response; in men, greater cortisol increase predicted less affiliation. The male pattern held across competitive outcomes — it was the magnitude of cortisol change, independent of whether the man won or lost, that predicted affiliative withdrawal.
Heinrichs et al. (2003) demonstrated that the presence of a trusted individual during psychosocial stress suppressed cortisol through the oxytocin system, and that the combination of social support and exogenous oxytocin produced the largest reduction in both cortisol and subjective anxiety. Follow-up work confirmed that OXTR genotype moderates this buffering capacity, and ecological momentary assessment studies have extended the finding from laboratory paradigms into daily life, where the pleasantness of social company tracks OXTR variation in its capacity to attenuate stress-related negative affect. In the absence of social support, exogenous oxytocin increased perceived social stress in men by amplifying self-referential processing of the social threat (Eckstein et al., 2014).
The two-channel structure of cortisol gating maps onto the behavioral space with a specific prediction. If elevated cortisol simultaneously suppresses testosterone-mediated competitive behavior (the dual-hormone finding) and independently suppresses affiliative behavior in men (the Sherman finding), then a man under sustained stress is pushed toward the bottom-left quadrant — withdrawn and disengaged — through parallel inhibition of both behavioral axes. The withdrawn state is an emergent property of simultaneous gating rather than a simple downstream consequence of competition-suppression.
From Architecture to Prediction
The developmental and adult evidence converge on a set of testable claims. Structured social inputs that provide physical co-presence, dyadic or group coordination, and tactile contact should produce measurable oxytocin responses; generic social contact — conversation without physical coordination, digital interaction without co-presence — should not. The removal of structured group inputs with bounded membership, positional differentiation, and productive orientation should produce endocrine signatures consistent with sustained cortisol gating: suppressed competitive engagement and suppressed affiliation simultaneously, the withdrawn profile rather than a simple shift along one axis. Individual variation in receptor sensitivity (AR CAG length, OXTR polymorphisms, MAOA activity) means the same structural input will produce different magnitudes of endocrine and behavioral response — a prediction that distinguishes the framework from purely sociological accounts, which would not expect within-population variation of this kind.
The father-son pathway adds a recursive prediction: populations in which paternal involvement has declined should show, one generation later, a shift in the distribution of affiliative baselines — more men entering the behavioral space with lower default oxytocin sensitivity, and therefore more men reaching the withdrawn quadrant under equivalent stress loads. This is a population-level prediction that requires longitudinal cohort data to test, and the endocrine measurement instruments to detect it exist. The following essay evaluates these and related predictions against naturalistic evidence from plant closures, marriage-market variation, sex-ratio perturbations, religious attendance patterns, and within-person endocrine measurement.
References
Auyeung, B., Lombardo, M. V., & Baron-Cohen, S. (2013). Prenatal and postnatal hormone effects on the human brain and cognition. Pflügers Archiv – European Journal of Physiology, 465(5), 557–571. https://doi.org/10.1007/s00424-013-1268-2
Bos, P. A., Panksepp, J., Bluthé, R.-M., & van Honk, J. (2012). Acute effects of steroid hormones and neuropeptides on human social-emotional behavior: A review of single administration studies. Frontiers in Neuroendocrinology, 33(1), 17–35. https://doi.org/10.1016/j.yfrne.2011.01.002
Bos, P. A., Terburg, D., & van Honk, J. (2010). Testosterone decreases trust in socially naïve humans. Proceedings of the National Academy of Sciences, 107(22), 9991–9995. https://doi.org/10.1073/pnas.0911700107
Burnstein, K. L., Maiorino, C. A., Dai, J. L., & Cameron, D. J. (1995). Androgen and glucocorticoid regulation of androgen receptor cDNA expression. Molecular and Cellular Endocrinology, 115(2), 177–186. https://doi.org/10.1016/0303-7207(95)03688-1
Butovskaya, M. L., Lazebny, O. E., Vasilyev, V. A., Dronova, D. A., Karelin, D. V., Mabulla, A. Z. P., Shibalev, D. V., & Ryskov, A. P. (2015). Androgen receptor gene polymorphism, aggression, and reproduction in Tanzanian foragers and pastoralists. PLOS ONE, 10(8), e0136208. https://doi.org/10.1371/journal.pone.0136208
Byrd, A. L., & Manuck, S. B. (2014). MAOA, childhood maltreatment, and antisocial behavior: Meta-analysis of a gene–environment interaction. Biological Psychiatry, 75(1), 9–17. https://doi.org/10.1016/j.biopsych.2013.05.004
Caspi, A., McClay, J., Moffitt, T. E., Mill, J., Martin, J., Craig, I. W., Taylor, A., & Poulton, R. (2002). Role of genotype in the cycle of violence in maltreated children. Science, 297(5582), 851–854. https://doi.org/10.1126/science.1072290
Crespi, B. J. (2016). Oxytocin, testosterone, and human social cognition. Biological Reviews, 91(2), 390–408. https://doi.org/10.1111/brv.12175
De Dreu, C. K. W., Greer, L. L., Handgraaf, M. J. J., Shalvi, S., Van Kleef, G. A., Baas, M., Ten Velden, F. S., Van Dijk, E., & Feith, S. W. W. (2010). The neuropeptide oxytocin regulates parochial altruism in intergroup conflict among humans. Science, 328(5984), 1408–1411. https://doi.org/10.1126/science.1189047
De Dreu, C. K. W., Greer, L. L., Van Kleef, G. A., Shalvi, S., & Handgraaf, M. J. J. (2011). Oxytocin promotes human ethnocentrism. Proceedings of the National Academy of Sciences, 108(4), 1262–1266. https://doi.org/10.1073/pnas.1015316108
De Dreu, C. K. W., & Kret, M. E. (2016). Oxytocin conditions intergroup relations through upregulated in-group empathy, cooperation, conformity, and defense. Biological Psychiatry, 79(3), 165–173. https://doi.org/10.1016/j.biopsych.2015.03.020
Dekkers, T. J., van Rentergem, J. A. A., Meijer, B., Popma, A., Wagemaker, E., & Huizenga, H. M. (2019). A meta-analytical evaluation of the dual-hormone hypothesis: Does cortisol moderate the relationship between testosterone and status, dominance, risk taking, aggression, and psychopathy? Neuroscience & Biobehavioral Reviews, 96, 250–271. https://doi.org/10.1016/j.neubiorev.2018.12.004
Diekhof, E. K., Wittmer, S., & Reimers, L. (2014). Does competition really bring out the worst? Testosterone, social distance and inter-male competition shape parochial altruism in human males. PLOS ONE, 9(7), e98977. https://doi.org/10.1371/journal.pone.0098977
Eckstein, M., Scheele, D., Weber, K., Stoffel-Wagner, B., Maier, W., & Hurlemann, R. (2014). Oxytocin facilitates the sensation of social stress. Human Brain Mapping, 35(9), 4741–4750. https://doi.org/10.1002/hbm.22508
Eisenegger, C., Haushofer, J., & Fehr, E. (2011). The role of testosterone in social interaction. Trends in Cognitive Sciences, 15(6), 263–271. https://doi.org/10.1016/j.tics.2011.04.008
Feldman, R., Gordon, I., Schneiderman, I., Weisman, O., & Zagoory-Sharon, O. (2010). Natural variations in maternal and paternal care are associated with systematic changes in oxytocin following parent-infant contact. Psychoneuroendocrinology, 35(8), 1133–1141. https://doi.org/10.1016/j.psyneuen.2010.01.013
Gettler, L. T., McDade, T. W., Feranil, A. B., & Kuzawa, C. W. (2011). Longitudinal evidence that fatherhood decreases testosterone in human males. Proceedings of the National Academy of Sciences, 108(39), 16194–16199. https://doi.org/10.1073/pnas.1105403108
Gleason, E. D., & Marler, C. A. (2013). Non-genomic transmission of paternal behaviour between fathers and sons in the monogamous and biparental California mouse. Proceedings of the Royal Society B: Biological Sciences, 280(1763), 20130824. https://doi.org/10.1098/rspb.2013.0824
Handa, R. J., & Weiser, M. J. (2014). Gonadal steroid hormones and the hypothalamo-pituitary-adrenal axis. Frontiers in Neuroendocrinology, 35(2), 197–220. https://doi.org/10.1016/j.yfrne.2013.11.001
Heinrichs, M., Baumgartner, T., Kirschbaum, C., & Ehlert, U. (2003). Social support and oxytocin interact to suppress cortisol and subjective responses to psychosocial stress. Biological Psychiatry, 54(12), 1389–1398. https://doi.org/10.1016/S0006-3223(03)00465-7
Hines, M. (2020). Neuroscience and sex/gender: Looking back and forward. Journal of Neuroscience, 40(1), 37–43. https://doi.org/10.1523/JNEUROSCI.0750-19.2019
Kirsch, P., Esslinger, C., Chen, Q., Mier, D., Lis, S., Siddhanti, S., Gruppe, H., Mattay, V. S., Gallhofer, B., & Meyer-Lindenberg, A. (2005). Oxytocin modulates neural circuitry for social cognition and fear in humans. Journal of Neuroscience, 25(49), 11489–11493. https://doi.org/10.1523/JNEUROSCI.3984-05.2005
Knight, E. L., Sarkar, A., Prasad, S., & Mehta, P. H. (2020). Beyond the challenge hypothesis: The emergence of the dual-hormone hypothesis and recommendations for future research. Hormones and Behavior, 123, 104657. https://doi.org/10.1016/j.yhbeh.2019.104657
Kolla, N. J., & Bortolato, M. (2020). The role of monoamine oxidase A in the neurobiology of aggressive, antisocial, and violent behavior: A tale of mice and men. Progress in Neurobiology, 194, 101875. https://doi.org/10.1016/j.pneurobio.2020.101875
Kosfeld, M., Heinrichs, M., Zak, P. J., Fischbacher, U., & Fehr, E. (2005). Oxytocin increases trust in humans. Nature, 435(7042), 673–676. https://doi.org/10.1038/nature03701
Kutlikova, H. H., Zhang, L., Eisenegger, C., van Honk, J., & Lamm, C. (2023). Testosterone eliminates strategic prosocial behavior through impacting choice consistency in healthy males. Neuropsychopharmacology, 48(11), 1541–1550. https://doi.org/10.1038/s41386-023-01570-y
Mascaro, J. S., Hackett, P. D., & Rilling, J. K. (2014). Differential neural responses to child and sexual stimuli in human fathers and non-fathers and their hormonal correlates. Psychoneuroendocrinology, 46, 153–163. https://doi.org/10.1016/j.psyneuen.2014.04.014
Mehta, P. H., & Josephs, R. A. (2010). Testosterone and cortisol jointly regulate dominance: Evidence for a dual-hormone hypothesis. Hormones and Behavior, 58(5), 898–906. https://doi.org/10.1016/j.yhbeh.2010.08.020
Nadler, A., Camerer, C. F., Zava, D. T., Ortiz, T. L., Watson, N. V., Carré, J. M., & Nave, G. (2019). Does testosterone impair men’s cognitive empathy? Evidence from two large-scale randomized controlled trials. Proceedings of the Royal Society B: Biological Sciences, 286(1910), 20191062. https://doi.org/10.1098/rspb.2019.1062
Ou, J., Wu, Y., Hu, Y., Gao, X., Li, H., & Tobler, P. N. (2021). Testosterone reduces generosity through cortical and subcortical mechanisms. Proceedings of the National Academy of Sciences, 118(28), e2021745118. https://doi.org/10.1073/pnas.2021745118
Plant, T. M., & Barker-Gibb, M. L. (2004). Neurobiological mechanisms of puberty in higher primates. Human Reproduction Update, 10(1), 67–77. https://doi.org/10.1093/humupd/dmh001
Sherman, G. D., Rice, L. K., Jin, E. S., Jones, A. C., & Josephs, R. A. (2017). Sex differences in cortisol’s regulation of affiliative behavior. Hormones and Behavior, 92, 20–28. https://doi.org/10.1016/j.yhbeh.2016.12.005
Simerly, R. B., Chang, C., Muramatsu, M., & Swanson, L. W. (1990). Distribution of androgen and estrogen receptor mRNA-containing cells in the rat brain: An in situ hybridization study. Journal of Comparative Neurology, 294(1), 76–95. https://doi.org/10.1002/cne.902940107
Terburg, D., & van Honk, J. (2013). Approach–avoidance versus dominance–submissiveness: A multilevel neural framework on how testosterone promotes social status. Emotion Review, 5(3), 296–302. https://doi.org/10.1177/1754073913477510
Valenzuela, N. T., Ruiz-Pérez, I., Rodríguez-Sickert, C., Polo, P., Muñoz-Reyes, J. A., Yeste-Lizán, A., & Pita, M. (2022). The relationship between androgen receptor gene polymorphism, aggression and social status in young men and women. Behavioral Sciences, 12(2), 42. https://doi.org/10.3390/bs12020042
van Wingen, G., Mattern, C., Verkes, R. J., Buitelaar, J., & Fernández, G. (2010). Testosterone reduces amygdala–orbitofrontal cortex coupling. Psychoneuroendocrinology, 35(1), 105–113. https://doi.org/10.1016/j.psyneuen.2009.09.007
Viau, V. (2002). Functional cross-talk between the hypothalamic-pituitary-gonadal and -adrenal axes. Journal of Neuroendocrinology, 14(6), 506–513. https://doi.org/10.1046/j.1365-2826.2002.00798.x
Weisman, O., Zagoory-Sharon, O., & Feldman, R. (2012). Oxytocin administration to parent enhances infant physiological and behavioral readiness for social engagement. Biological Psychiatry, 72(12), 982–989. https://doi.org/10.1016/j.biopsych.2012.06.011
Wilson, C. A., & Davies, D. C. (2007). The control of sexual differentiation of the reproductive system and brain. Reproduction, 133(2), 331–359. https://doi.org/10.1530/REP-06-0078